IVERMECTIN's Little Secret
Ivermectin May Help Prevent SARS-CoV-2 Conversion of M1 LB- Foamy Macrophages (Protector) to M2 LB+ Foamy Macrophages (Non-protector) by Preventing MAPK8/Jnk1 Phosphorylation
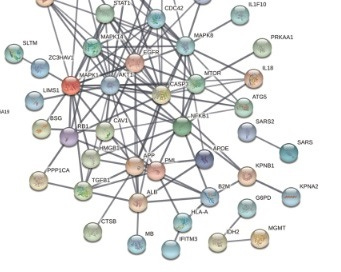
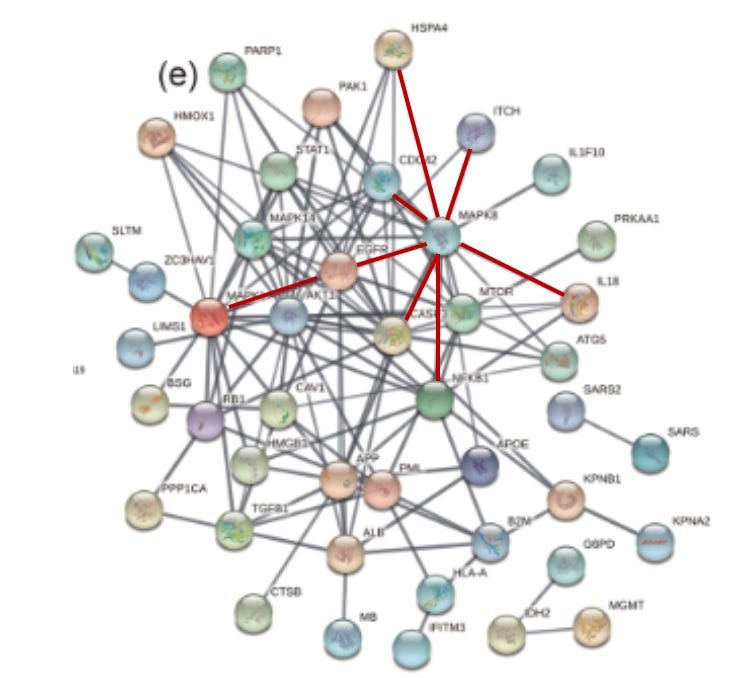
Figure 1. The Ivermectin - SARS-CoV-2 String PPI Network Map
Li N et al., 2021.
In an effort to understand how ivermectin might protect against COVID-19 severity, Li N et al., 2021 derived a 52 protein-protein interaction (PPI) string network map (Figure 1) reflecting proteins affected by ivermectin in an ovarian cancer cell line and which had previously identified to directly interact with SARS-CoV-2. Notable proteins identified include BSG (foam cell formation), EGFR (foam cell formation), CASP3 (apoptosis), NFKB1 (induction of pro-inflammatory mediators), IFITM3 (interferon stimulated antiviral gene) and TGFB1 (may bind and activate AFP). It is argued elsewhere (Laderoute M., Open Heart, April 26, 2021) that AFP (alpha-fetoprotein) was mislabeled as ALB in the PPI string network. Albumin is a protein highly related to AFP but is involved in maintaining osmotic pressure in the blood and is responsible for the transport of various chemicals. This confusion may have been because the 8 peptide sequences did not distinguish AFP from ALB and ovarian cancer cells do not express ALB but express AFP. Unlike ALB, AFP plays a well established role in virulence and in malignant potential, and is the central modulator of immunosenescence. Note that TP53 (downmodulates AFP expression) was not captured in the PPI as ovarian cancer cell lines generally do not express functional TP53.
The implicated proteins and pathways were recognized to play an important role in the generation of LB- foamy macrophages (see Figure 2) involving BSG and EGFR in foam cell formation which involves the production of HERV-K102 particles. BSG is a master regulator of foam cell formation (Lv JJ et al., 2021). Additionally EGFR regulates foam cell formation in macrophages (Orekhov AN et al., 2018) and in sebocytes (Zhang MF et al., 2018).
It should be noted that diseases associated with BSG include Severe Acute Respiratory Syndrome (https://www.genecards.org/cgi-bin/carddisp.pl?gene=BSG). As well a monoclonal antibody to BSG partially inhibits SARS-CoV-2 replication in a human BSG transgenic mouse model and blocks cytokine storm (Geng et al, 2021). Thus, the Ivermectin - SARS-CoV-2 String PPI Network Map identified by Li N et al, 2021 is clearly relevant to COVID-19 pathogenesis.
Foam cell formation in LB- foamy macrophages (Figure 2) and in sebocytes (Figure 3) involves HERV-K102 expression (Laderoute et al, 2007, 2015; Nelson AM et al, 2008) where the production of particles creates vacuoles giving the cells a lacey appearance. These foam cells are easily distinguished from the LB+ foamy macrophages (anti-inflammatory foamy macrophages) which differ from the LB- foamy macrophages as the former stain positive for Oil Red O, a stain for cholesterol esters stored as lipid bodies in the cytoplasm (Figure 2).
As recently reviewed elsewhere, the HERV-K102 particle system appears to play a critical role in trained innate immunity defense especially against emerging RNA viruses (Laderoute M. hervk102@substack.com, January 24, 2022). Therefore this validated the clinical significance of using ivermectin for the early treatment of COVID-19 patients to restore innate immunity and circumvent COVID-19 severity.
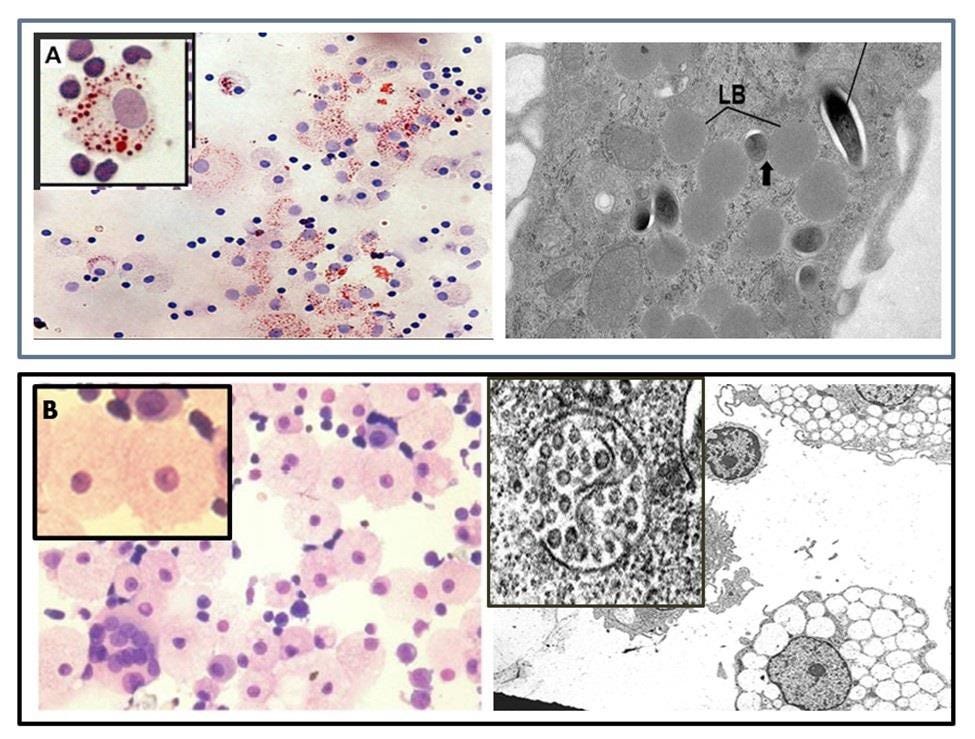
Figure 2. Two Types of Foamy Macrophages (FM).
A) Oil Red Oil positive, lipid body positive (LB+ FM) which are considered M2 (anti-inflammatory) and the
B) Oil Red O negative, lipid body negative (LB- FM) which are highly vacuolated and produce HERV-K102 particles and are considered M1-like (proinflammatory).
The LB- FMs may provide trained innate immunity against emerging RNA viruses. A) From Peyron P et al., PLoS Path, 2008 and B) from Laderoute MP. Discovery Medicine, 2015, Laderoute M et al., AIDS 2007, and Laderoute M et al., Open AIDS J, 2015.
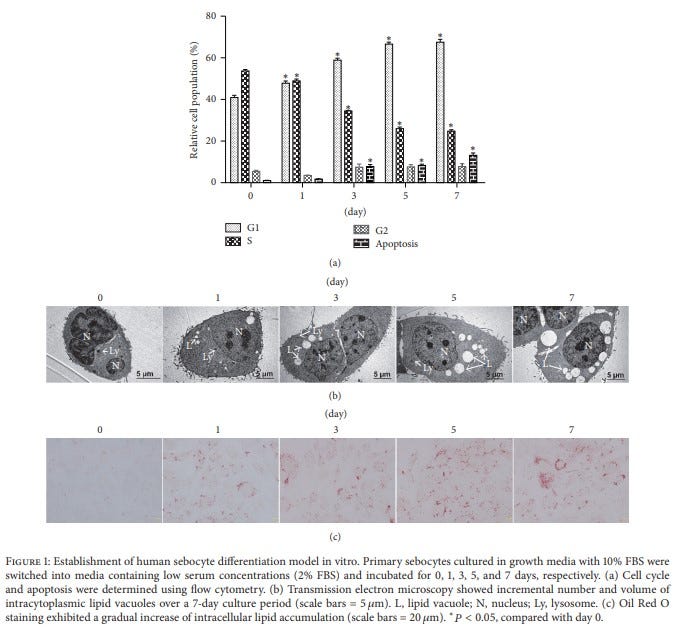
Sebocytes are specialized foamy macrophages in sebaceous glands that line the mucosa. They constitutively produce HERV-K102 particles and like macrophages, release them by lysis around day 7. The waxy sebum containing the particles exits the gland and coats the mucosa, thus providing humans with the first line of defense against emerging RNA viruses. As shown in Figure 3 (Zhang MF et al., 2018), during the 7 days of differentiation, primary sebocytes develop the unique morphology (highly vacuolated) of the LB- foamy macrophages (b) and do not significantly stain with Oil Red Oil (‘c’). Like the LB- foamy macrophages, they are programmed for cell death on day 7 (a).
Figure 3. Primary sebocytes differentiating over 7 days develop the morphology of the LB- foamy macrophages (b) , do not stain well with Oil Red Oil (‘c’) and undergo apoptosis on day 7 (a). (Zhang et al., 2018).
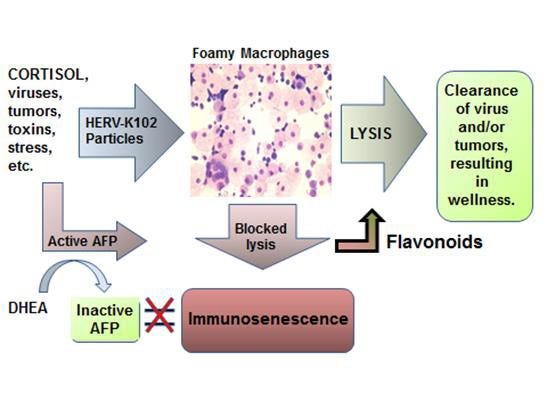
The ivermectin-SARS-CoV-2 PPI network was interpreted to mean that ivermectin reverses immunosenescence (Laderoute M., Open Heart, April 26, 2021). Immunosenescence refers to the failed lytic release of HERV-K102 particles from LB- foamy macrophages (Figure 4).
Figure 4. The New Immunosenescence Paradigm, 2015 (Laderoute, 2015)
Immunosenescence refers to the failed lytic release of HERV-K102 particles from foamy macrophages. Zinc, DHEA and flavonoids (eg. 50 mg of genistein) appear to bind and inactivate AFP, reversing immunosenescence. Ivermectin may reverse immunosenescence although it is not known if it binds and inactivates AFP.
Interestingly, various lines of evidence implicates SARS-CoV-2 mediated immunosenescence associated with severe COVID-19 (Laderoute M. hervk102@substack.com, January 27, 2022). For example, Liao M et al, 2020 showed the macrophages in bronchoalveolar lavage fluids (BALF) from severe COVID-19 patients exhibited apoptosis resistance associated with a malignant phenotype.
However, there have been many reports that SARS-CoV-2 does not infect monocytes/macrophages due to the lack of expression of ACE2 and TMPRSS2.
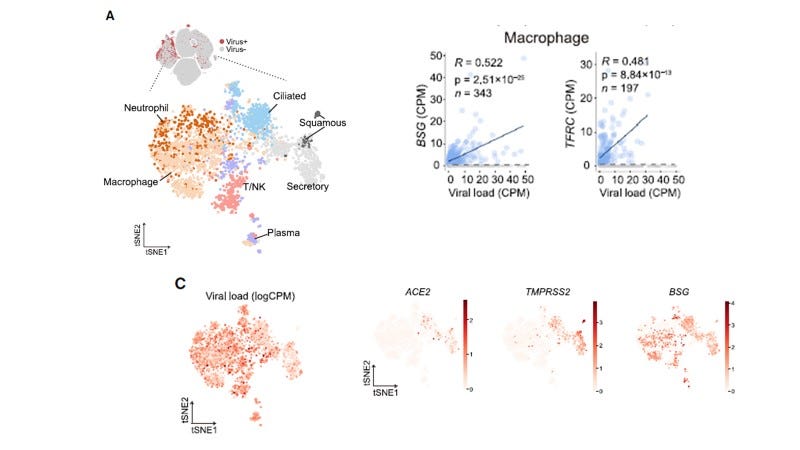
Nevertheless Ren X et al, 2021 reported in the BALF from severe COVID-19 patients, although the macrophages did not express ACE2 or TMPRSS2, they had high levels of SARS-CoV-2 RNA (Figure 5) where both positive and negative strands were detected showing SARS-CoV-2 was replicating in these cells.
Figure 5. Macrophages from BALF of Severe COVID-19 Patients Contain Higher Levels of SARS-CoV-2 RNA than Epithelial Cells [From Ren X et al., Cell, 2021 Figure 4.]
SARS-CoV-2 RNA was not detected in macrophages from BALF samples from moderate patients, and was never detected in peripheral blood mononuclear cells (PBMC) from any COVID-19 patient despite the expression of BSG in monocytes/macrophages generally. BSG and the transferrin receptor (TFRC) levels correlated with viral load in macrophages from the severe COVID-19 BALF patients (Figure 5) consistent with upregulation following SARS-CoV-2 infection. Both BSG and TFRC have been reported as co-receptors for SARS-CoV-2 entry although this has been disputed.
Although Ren et al did not discuss the issue of Antibody Dependent Enhancement (ADE) of infection, he showed that COVID-19 severity was strongly correlated with enrichment of the B_C05-MZB1-XBP1 plasma B cell subset which produced primarily IgA1 and IgG1 antibodies. These cells contained sequences known to be common to SARS-CoV-2 neutralizing antibodies (NAbs). With convalescence in the severe COVID-19 patients, this plasma B cell subset diminished. Thus, it is likely ADE enhanced the infection of the BSG positive macrophages but only in patients with severe COVID-19 (ie. patients who developed the suboptimal neutralizing antibodies). This explains how macrophages are resistant to infection by SARS-CoV-2 but in severe cases of COVID-19, SARS-CoV-2 infects macrophages.
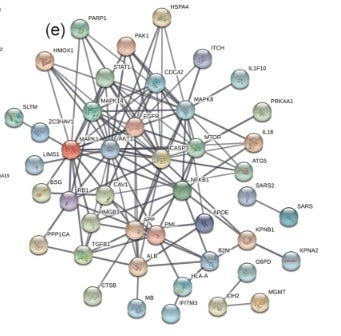
Ren et al., listed the differentially expressed genes (DEGs) that were upregulated in the SARS-CoV-2 infected macrophages when compared to the uninfected. The ones that were in the ivermectin-SARS-CoV-2 PPI network were; MAPK1, AKT1, CASP3, NFKB1, HSPA4, CDC42, IL18 and ITCH. These were overlaid onto the PPI network (Figure 6). Although MAPK8 (JNK1) was not identified as a DEG (activation involves phosphorylation of the protein which would not be identified by increases in mRNA), all the affected DEGS except AKT1, identified MAPK8 (JNK1) as the hub. (AKT1 indirectly connected to MAPK8 via CASP3, MAPK1 or HSPA4).
Figure 6. DEGS Identified to be Upregulated in SARS-COV-2 Infected Macrophages Over Uninfected Macrophages in BALF from Severe COVID-19 Patients Center on MAPK8/JNK1 in the Ivermectin-SARS-CoV-2 PPI String Network
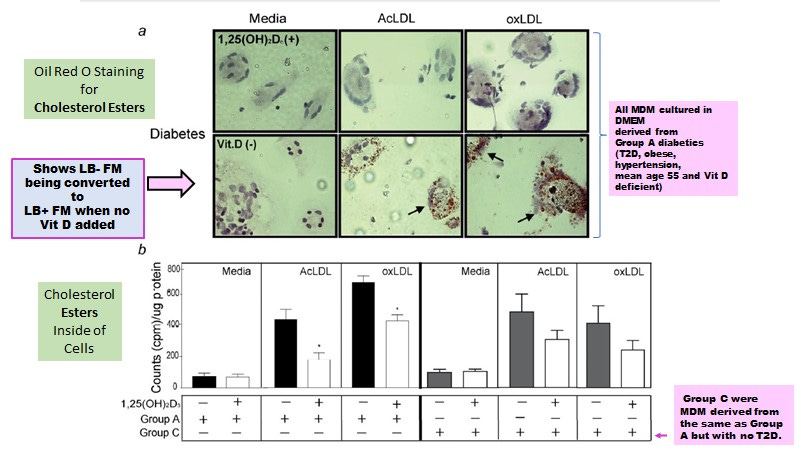
Coincidently while searching for a role of Vitamin D deficiency in terms of affects on foam cell formation, it was discovered that Vitamin D deficiency was associated with an enhanced conversion of the LB- FMs to the LB+ FMs (Figure 7a) exacerbated in those with hyperglycemia and/or insulin resistance (T2D) (Figure 7b). Note that the addition of LDL promotes the differentiation of M0 macrophages to M1 LB- FM in the presence of Vitamin D while in its absence, LDL promotes the differentiation of M0 macrophages to M2 LB+ FM (Figure 7).
Figure 7. Vitamin D Blocks the Conversion of LB- Foamy Macrophages to LB+ Foamy Macrophages (from Oh J et al, 2009)
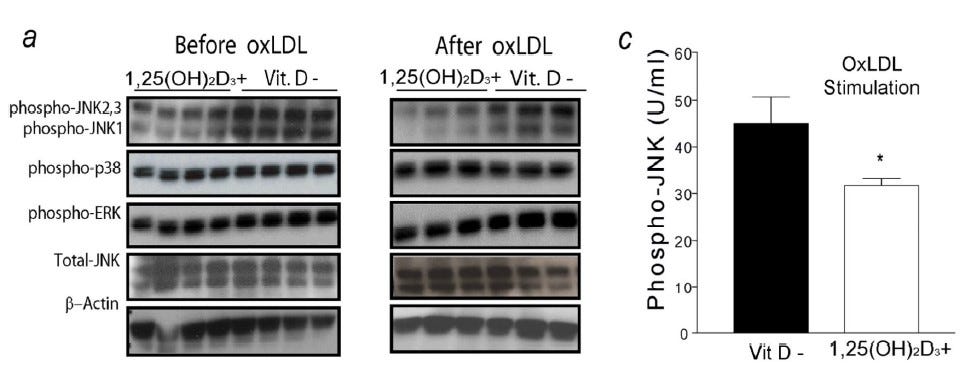
The absence of Vitamin D favors the phosphorylation and activation (data not shown) of JNK1 (MAPK8) (Figure 8) at either the MO stage or after differentiation with LDL.
Figure 8. Sufficient Vitamin D blocks JNK1 Phosphorylation in M0 Macrophages and in M0 Differentiated to Become FM (from Oh J et al, 2009)
So, in addition to Vitamin D dampening abnormal TLR or NFKB1 signaling and balancing the Th1 to Th2 responses favoring Th1 which may attenuate cytokine storm in severe COVID-19 patients (Figure 9), it also balances the FM responses favoring M1 (LB- FM) over M2 (LB+ FM). This protects trained innate immunity needed for defense against emerging RNA viruses by enhancing the HERV-K102 particle defense system of the LB- FMs.
Figure 9. Summary of Known Effects of Sufficient Versus Insufficient Vitamin D Levels in COVID-19 Pathogenesis
(from Bae JH et al., 2022).
So in summary, it appears that SARS-CoV-2 infection occurs in macrophages but only in the BALF of severe cases, most likely related to ADE. This infection may be associated with the activation of MAPK8/JNK1 by phosphorylation which may convert M1 pro-inflammatory LB- foamy macrophages to non-inflammatory M2 LB+ foamy macrophages, abrogating HERV-K102 trained innate immunity. It is possible that ivermectin like Vitamin D, may help overcome the phosphorylation of JNK1 by SARS-CoV-2. Thus, ivermectin may not only reverse immunosenescence in people at increased risk of COVID-19 severity, but may also prevent conversion of LB- FMs to LB+ FMs by SARS-CoV-2. This may help explain why ivermectin seems to synergize with Vitamin D, zinc and flavonoids in attenuating COVID-19 severity. This is ivermectin’s little secret mode of action.
REFERENCES
Bae JH, Choe HJ, Holick MF, Lim S. Association of vitamin D status with COVID-19 and its severity : Vitamin D and COVID-19: a narrative review. Rev Endocr Metab Disord. 2022 Jan 4:1–21. doi: 10.1007/s11154-021-09705-6. Epub ahead of print. PMID: 34982377; PMCID: PMC8724612.
Geng J, Chen L, Yuan Y, et al . CD147 antibody specifically and effectively inhibits infection and cytokine storm of SARS-CoV-2 and its variants delta, alpha, beta, and gamma. Signal Transduct Target Ther. 2021 Sep 25;6(1):347. doi: 10.1038/s41392-021-00760-8.
Laderoute MP, Giulivi A, Larocque L, et al. The replicative activity of human endogenous retrovirus K102 (HERV-K102) with HIV viremia. AIDS. 2007 Nov 30;21(18):2417-24.
Laderoute MP, Larocque LJ, Giulivi A, et al. Evidence that human endogenous retrovirus K102 is a replication competent foamy virus that may antagonize HIV-1 replication. Open AIDS J. 2015 Dec 7;9:112-22. doi: 10.2174/1874613601509010112.
Laderoute MP. A new paradigm about HERV-K102 particle production and blocked release to explain cortisol mediated immunosenescence and age-associated risk of chronic disease. Discov Med. 2015 Dec;20(112):379-91.
Laderoute M. Ivermectin May Prevent and Reverse Immunosenescence By Antagonizing Alpha-fetoprotein and Downmodulating PI3K/Akt/mTOR Hyperactivity. Open Heart April 29, 2021. https://openheart.bmj.com/content/8/1/e001655.responses#ivermectin-may-prevent-and-reverse-immunosenescence-by-antagonizing-alpha-fetoprotein-and-downmodulating-pi3k-akt-mtor-hyperactivity.
Laderoute M. What you need to know about the HERV-K102 innate immunity protector system of macrophages against RNA pandemic viruses. hervk@substack.com, January 26, 2022.
Laderoute M. Part II: How SARS-CoV-2 Targets HERV-K102 in Sebocytes and Foamy Macrophages. hervk@substack.com, January 28, 2022.
Li N, Zhao L, Zhan X. Quantitative proteomics reveals a broad-spectrum antiviral property of ivermectin, benefiting for COVID-19 treatment. J Cell Physiol. 2021;236(4):2959-2975. doi:10.1002/jcp.30055.
Liao M, Liu Y, Yuan J, et al. Single-cell landscape of bronchoalveolar immune cells in patients with COVID-19. Nat Med. 2020 Jun;26(6):842-844. doi: 10.1038/s41591-020-0901-9.
Lv JJ, Wang H, Cui H-Y et al. Blockade of macrophage CD147 protects against foam cell formation in atherosclerosis. Front Cell Dev Biol 2021. doi.org/10.3389/fcell.2020.609090.
Nelson AM, Zhao W, Gilliland KL, Zaenglein AL, Liu W, Thiboutot DM. Neutrophil gelatinase-associated lipocalin mediates 13-cis retinoic acid-induced apoptosis of human sebaceous gland cells. J Clin Invest. 2008 Apr;118(4):1468-78. doi: 10.1172/JCI33869.
Oh J, Weng S, Felton SK et al. 1,25(OH)2 vitamin D inhibits foam cell formation and suppresses macrophage cholesterol uptake in patients with type 2 diabetes mellitus. Circulation. 2009 Aug 25;120(8):687-98. doi: 10.1161/CIRCULATIONAHA.109.856070.
Orekhov AN, Oishi Y, Nikiforov NG, Zhelankin AV, Dubrovsky L, Sobenin IA, Kel A, Stelmashenko D, Makeev VJ, Foxx K, Jin X, Kruth HS, Bukrinsky M. Modified LDL Particles Activate Inflammatory Pathways in Monocyte-derived Macrophages: Transcriptome Analysis. Curr Pharm Des. 2018;24(26):3143-3151. doi: 10.2174/1381612824666180911120039.
Peyron P, Vaubourgeix J, Poquet Y, et al. Foamy macrophages from tuberculous patients' granulomas constitute a nutrient-rich reservoir for M. tuberculosis persistence. PLoS Pathog. 2008 Nov;4(11):e1000204. doi: 10.1371/journal.ppat.1000204.
Ren X, Wen W, Fan X, et al. COVID-19 immune features revealed by a large-scale single-cell transcriptome atlas. Cell. 2021;184(7):1895-1913.e19. doi:10.1016/j.cell.2021.01.053.
Zhang MF, Cai XL, Jing KP, Pi XX, Liao PY, Li SJ, Wen-Li, Cai CC, Quan JH, Fan YM, Ge-Shi. Differentiation Model Establishment and Differentiation-Related Protein Screening in Primary Cultured Human Sebocytes. Biomed Res Int. 2018 Apr 5;2018:7174561. doi: 10.1155/2018/7174561.




It should be noted that high viral loads in the nasopharyngeal swabs (NPS) correlated with the presence of SARS-CoV-2 spike protein specific Abs/NAbs in serum while low viral loads were correlated with the absence of antibody (Liu W et al. Emerging Infectious Diseases, 2021). This showed ADE likely played a role in infection.
In Figure 5 f and g of the Huang N et al Nature Medicine, May 2021 paper they demonstrate SARS-CoV-2 protein and mRNA in sebocytes isolated from saliva of a mild COVID-19 patient (see image showing this in a previous blog on how SARS-CoV-2 targets HERV-K102). First, there should NOT be any intact sebocytes in saliva from normal healthy adults as they all lyse prior to being exuded from the glands as sebum. Huang et al did not address this but the lack of intact sebocytes being isolated from normal healthy adults in saliva and used as a negative control probably reflects there weren't any. The patient probably had suboptimal neutralizing antibodies due to vaccination or a previous moderate to severe infection which mediated ADE for entry into sebocytes. Presumably natural immunity including T cell responses and antibodies to other proteins kept the infection mild. The failure of sebocytes to lyse upon SARS-CoV-2 infection substantiates the notion that SARS-CoV-2 infection prevents apoptosis a key feature of immunosenescence.
One dose of the mRNA vaccines (which induces innate immunity) in the USA was associated with the lowest viral loads in NPS stratified by vaccination status in May 2021 (for 2 of 3 SC2 targets) before the emergence of the delta variant. After delta became dominant by July 2021, there was no difference in NPS viral loads by vaccination status and where all showed higher viral loads than in May (Griffin JB et al., MMWR, 70: 1170-1176, 2021). Two things may be happening here. For the 2 dose vaccinated ADE may be promoting infection of sebocytes in May 2021 whereas innate immunity provided some protection. The delta variant might have less affinity for the ACE2 entry receptor and higher affinity for BSG, meaning this variant could equally enter the sebocytes regardless of the presence or not of suboptimal neutralizing antibodies.
The Ren et al Cell, 2021 study was conducted before the emergence of delta. So it could mean that with the delta variant, more macrophages in BALF actually used BSG to enter cells in addition to enhancement through CD32 (FCGR2a) which by the way gets upregulated with monocyte to macrophage differentiation. So it will be important to see if the issue of phosphorylation of MAPK8/JNK1 persists when BSG is preferentially used to enter macrophages. Note this helps to also explain the controversy as to whether BSG is used for SARS-CoV-2 entry in the absence of ADE. However, it does complicate the question of how ivermectin prevents COVID-19 severity.
World
I talked to a bomber pilot a couple of years ago
shawn
21 min ago
A very interesting gentleman, affected by the war ww2.
The great backpeddle is starting along with the great retrenching and refortifying. Luckily for the planet there are honourable scientist doctor researchers in the world. people that stood against the corporate profiteers. We are greatful.
As we know now the corporate machine of distraction is spinning up and I think working well.
We must not be distracted with all the superfluous studies the if / ands /or whats.
Does Ivermectin work, does Hydroxy work, were there protocols available to treat. When were the WHO/CDC/FDA made aware.
If the answer is yes. If those treatments/ prophylaxis were suppressed then murder has been committed. Nothing else matters right now. We must prove our case. Period.
Who committed those murders
Who participated in the great lie, news media of course, doctors, legislators etc…
How do we make people accountable, there is a way that won’t destroy society. I will offer my suggestion in another posting.
I asked that bomber pilot about flak, he said it was somewhat effective. A plane would fly into a cloud of flak which led to the occasional mission failure. The most common thing flak did was to distract and force the plane of course.
Lets not let the flak distract or force us off course.
Is Ivermectin effective, does Hydroxy work, where treatment options available, when did they know.
Shawn663