What you need to know about the HERV-K102 innate immunity protector system of macrophages against RNA pandemic viruses
For Future Pandemic Preparedness
January 26, 2022
PART I: AN INTRODUCTION TO HERV-K102: A PROTECTOR FOAMY (SPUMA) RETROVIRUS OF HUMANS
It is NOT widely appreciated that humans have a unique innate protector system against emerging epidemic or pandemic RNA viruses which involves a ‘virus anti-virus’ response. Much like the old adage ‘to fight fire with fire’, it seems less than 1 million years ago humans acquired an endogenous foamy retrovirus called human endogenous retrovirus K102 (HERV-K102) which helps protect against emerging (pandemic) RNA viruses, such as HIV-1.
HERV-K102 particle production has been described in cord blood mononuclear cells (CB) when cultured in IMDM but not RPMI media (Figure 1). Most immunology research employs RPMI media which appears to block macrophage foam cell formation. This helps explain why little is known about HERV-K102 particle production and why it is often missed. As shown in Figure 1, during culture of CB in IMDM media, macrophages accumulate HERV-K102 particles in vacuoles, giving the cells a foamy appearance. This is why this type of retrovirus is called a foamy virus.
Figure 1. Foamy Macrophages Accumulate HERV-K102 Particles.
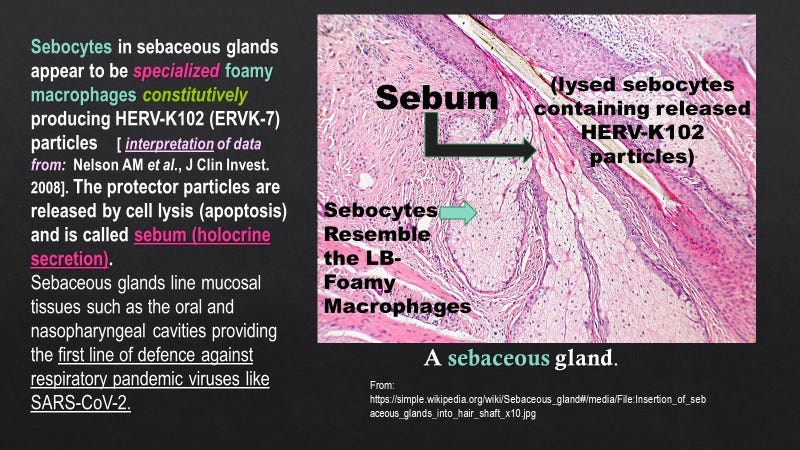
Foamy or spuma retroviruses are well established to be non-pathogenic, have been shown in all species examined, and are known to co-evolve with the host, suggesting a symbiotic relationship. Nevertheless, their role in health or disease remains a mystery. They are easily transmissible via biting and are known to replicate in sebocytes of sebaceous glands which line the oral cavity. HERV-K102 expression has also been demonstrated in sebocytes which, incidentally, morphologically resemble foamy macrophages, carry all the markers of foamy macrophages and moreover behave as functional macrophages. It is important to appreciate that sebocytes like foamy macrophages undergo programmed cell death around day 7 releasing the precious protector HERV-K102 particles via lysis. This results in what is known as holocrine secretion, where the waxy ‘sebum’ is released to the lumen and thus, the exterior of the body within mucosal tissues (Figure 2). In this manner, protector HERV-K102 constitutively replicates in a specialized niche without systemically disrupting the immune system and at the same time, provides the first line of defense especially against emerging RNA viruses at the mucosal interface.
Figure 2. Sebocytes Resemble Foamy Macrophages and Release HERV-K102 Particles as Sebum to Coat Mucosal Tissues (First Line of Protection)
While non-pathogenic in nature, foamy retroviruses have been shown to undergo lytic infection in some fibroblast cell lines but not others.
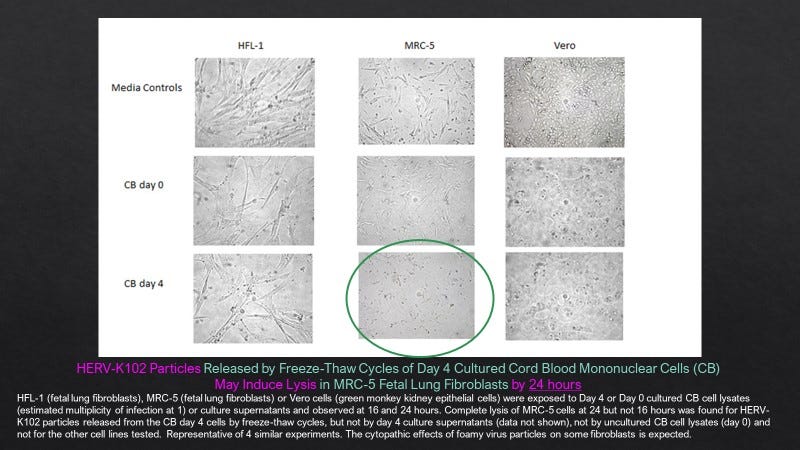
Not surprisingly, as expected for a foamy retrovirus, HERV-K102 particles obtained from cultured cord blood mononuclear cells (CB) also undergoes lytic infection in some but not all fibroblast cell lines. With an estimated multiplicity of infection of one (MOI=1), particles released from cells by repeated freeze-thaw cycles (but not culture supernatants) caused complete lysis of MRC-5 fibroblasts but not HFL-1 fibroblast cells at a very rapid, 24 hours (Figure 3). This would be expected for a fully functional and replication competent foamy retrovirus. It should be noted that by electron microscopy, HERV-K102 particles do not bud through the membrane like many other viruses but instead are released by a specialized apoptosis (lytic) process where the initiating signal is cytoplasmic (see notes in Figure 1). Presumably this would reflect when sufficient numbers of particles are produced in a given cell. Supernatants of cultured CB do not cause lysis of MRC-5 cells confirming the electron microscopy observations that the particles do not bud from CB cells.
Figure 3. HERV-K102 Particles Released from Cultured Cord Blood Mononuclear Cells (CB) Rapidly Induce Lysis in MRC-5 but not HFL-1 Fetal Lung Fibroblasts.
Nevertheless, despite the mystery concerning the function of foamy viruses generally, in humans it has been shown that HERV-K102 proteins (also HERV-K HML-2 proteins), particles or innate T cells and B cells recognizing HERV-K102 envelope protect against HIV-1 and breast cancers (detailed in Laderoute M, 2018: https://doi.org/10.12688/f1000research.11818.2). Evidently, cells which become transformed by viruses or tumor cells express HERV-K102 envelope on their surface but is not found on normal healthy cells. This cell surface expression can be recognized by antibody or T cells to the envelope protein and allows for clearance of these abnormal cells by antibody or the innate T cells that have specificity for envelope.
Indeed, there is some evidence to suggest that the expression of HERV-K102 envelope in cells contributes to the transformation process. Be that as it may, apparently, triggering of HERV-K102 envelope such as by an antibody directly and rapidly induces a cell death signal as has been shown in breast cancer cells both in vitro and in vivo (the latter in xenograft models). It is known that transformation induces a sensitivity to apoptosis induction and it is possible that HERV-K102 envelope along with antibody exploits this pathway for rapid clearance of virus infected and/or tumor cells.
Aside from the data in Figure 3 showing HERV-K102 particles can rapidly undergo selective lytic infection, the protector roles of the HERV-K102 particles themselves has not been well studied. We know that the particles are released from foamy macrophages/sebocytes by lysis on day 7 which in itself could offer protection against infection of foamy macrophages or sebocytes by external viruses (ie., lytic release of HERV-K102 particles kills the cells co-infected by a pathogenic virus). It remains to be addressed if the selectivity for a lytic infection by HERV-K102 is for cells transformed by viruses, those infected with other intracellular pathogens and/or in cells that are tumor transformed. However, we know in normal cells, HERV-K102 as a retrovirus, simply integrates into genomic DNA which would increase its genomic copy number and which may thus, amplify the protector response when challenged at a later time. Foamy retrovirus enhancement of gene copy number has been shown to be as high as 20 fold in hematopoietic tissues. Thus, the HERV-K102 protector system although pertains to innate immunity, can be amplified rivalling adaptive immunity mechanisms. This is a novel accomplishment for innate protector systems. Plus its induction can be very fast from zero particles per ml of plasma to 2.55 x 10 (11) in vivo in a matter of 84 hours.
In a future post we will talk about how HERV-K102 may be a primary mediator of trained innate immunity (a memory response of innate immunity providing enhanced heterologous protection against all kinds of intracellular pathogens and tumors).
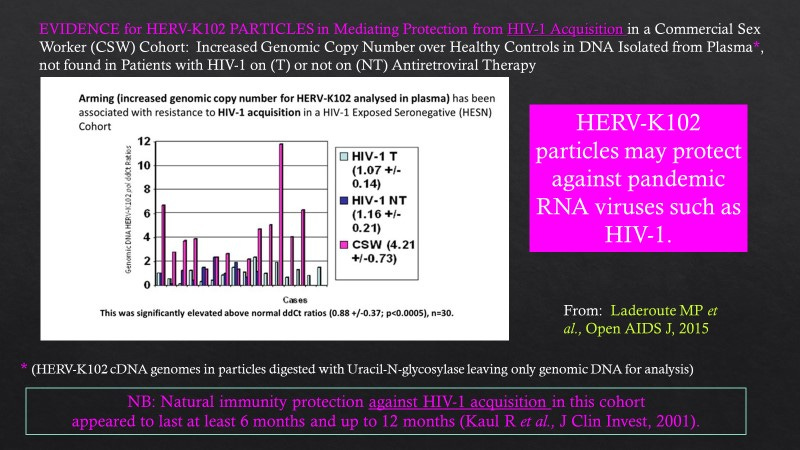
In terms of increased copy number of the pro-virus HERV-K102, there are data which suggests that HERV-K102 activity at least involving replication of HERV-K102 particles may provide sterilizing protection against HIV-1 acquisition (Figure 4).
Figure 4. Evidence for Protection Against Acquisition in an HIV-1 Exposed Seronegative (HESN) Cohort
Here (Figure 4) we see that protection against HIV-1 acquisition in an HIV-1 Exposed Seronegative (HESN) cohort is associated with an almost 5-fold increase in HERV-K102 genomic copy number (performed on genomic DNA sloughed into plasma) over normal healthy controls and which was not found in individuals infected with HIV-1 irrespective of use of anti-retrovirals.
These findings are highly clinically significant.
First, this data confirms the replication competence of HERV-K102 as was seen in vitro when CB was cultured in IMDM but not RPMI media (Figure 1), and as detected in vivo in patients infected with various viruses (Figure 5). Using GEO Profiles (https://www.ncbi.nlm.nih.gov/geoprofiles/) and searching for ERVK-7 as the gene symbol for HERV-K102, others have demonstrated monocytes, macrophages, alveolar macrophages and leukocytes express HERV-K102 while other types of cells infected with herpes viruses also express HERV-K102 sequences. Many viruses are known to transactivate HERV-K HML-2 sequences. Several research groups have confirmed that HERV-K102, is induced by HIV-1 in vitro (Brinzevich et al., 2014; Vincendeau et al., 2015). Moreover, HERV-K102 may be the only full length, human specific, HML-2 element induced by HIV-1 and/ or Tat (Gonzalez-Hernandez et al., 2012).
Figure 5. HERV-K102 Particles [Up to 10(12) per ml of Plasma] or Antibody to HERV-K102 Envelope Are Detected in Patients with Viremia.
Note: Despite the notable inhibition of HERV-K102 particle production in patients with HIV-1, over 96 % of patients with HIV-1 had detectable serum antibody to HERV-K102 envelope and/or demonstrable particles in plasma. This showed that the HERV-K102 protective system was a universal response to a pandemic RNA virus in humans.
Indeed, HERV-K102 envelope specific antibodies have been reported by Wang-Johanning’s research team in patients with various cancers over the years (since 2002) and as shown here for viral infections. As well, elevated antibodies recognizing HERV-K102 envelope peptide sequences have also been demonstrated in patients with amyotrophic lateral sclerosis (ALS) but correlations with antibodies to TPD-43 (the latter which upregulates HERV-K LTRs) were only detected in ALS patients (Simula ER et al., Viruses, 2021). Antibodies to HERV-K envelope have been reported more generally in various autoimmune diseases but where the peptide specificity had not been addressed. Recall that in peripheral blood mononuclear cells of humans, of HERV-Ks, only HERV-K102 and HERV-K18 produce envelope protein (Brinzevich D et al., J. Virol, 2014). Thus, these antibodies are suggestive of HERV-K102 and/or K18 envelope expression.
Second, for the first time this data shows that HERV-K102 replication (enhanced gene copy number) may be associated with sterilizing immunity (HIV-1 exposed but seronegative) and…. against a pandemic RNA virus, HIV-1.
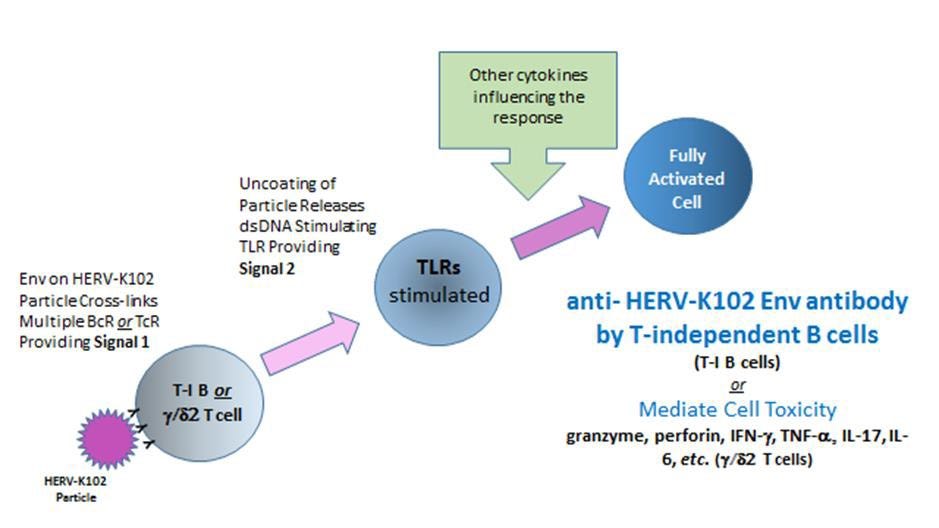
However, technically, this data does not distinguish the role of HERV-K102 particles themselves from the role of replicating particles in the generation of innate T cells or antibody to HERV-K102 envelope or other mechanisms in providing sterilizing immunity. For example, while expression of HERV-K102 proteins during its replication may render HIV-1 particles non-infectious [1], it is likely that only the antibody to HERV-K102 envelope would clear HIV-1 particles. For example, HIV-1 particles can be pseudotyped by a related HERV-K18 envelope (also located on chromosome 1 and induced by HIV-1), although not by HERV-K102 envelope (Brinzevich D et al., J. Virol, 2014). On the other hand, when HIV-1 buds through the cell surface it may carry HERV-K102 envelope in the lipid bilayer which surrounds the egressed HIV-1 particles as HIV-1, like SARS-CoV-2, is an enveloped RNA virus. In all likelihood, the HERV-K102 proteins, particles and the innate T and B cell responses all contribute to sterilizing immunity against HIV-1, a pandemic RNA virus. That the HERV-K102 particles themselves may induce or trigger the innate T and B cell response to HERV-K102 envelope has been hypothesized as explained in Figure 6.
_________________________________________________________________________
[1] For example, the HERV-K HML-2 protease cuts HIV-1 proteins in the wrong places abrogating HIV-1 infectivity.
__________________________________________________________________________
Figure 6. Hypothesis of How HERV-K102 Particles May Trigger Innate T and B Cells with Specificity for HERV-K102 Envelope (Env)
Phylogenetic Evidence Supporting HERV-K102 in Defending Against RNA Viruses
It should be noted that foamy retroviruses have cDNA genomes and thus a reversed lifecycle to pathogenic retroviruses (called orthoretroviruses) which instead have RNA genomes. Orthoretroviruses reverse transcribe upon entry into cells, whereas foamy retroviruses reverse transcribe upon exit from cells. This difference in part may account for the more stable genomes of foamy retroviruses and is consistent with the notion that foamy retroviruses may have co-evolved in the host to deal with orthoretroviral invasion as they would be able to replicate earlier than orthoretroviruses due to their cDNA genomes.
This seems to be relevant for the HERV-K HML-2 lineage of HERV-K102.
In this regard, accumulating phylogenetic evidence is consistent with a potential role of HERV-K HML-2 in limiting invasion by orthoretroviruses (Magiorkinis et al., 2015). Ancestral HML-2 elements emerged about 10.3 million years ago (mya) (Subramanian et al., 2011). There has been a striking decline of insertions of ERVs in the last 10 million years (My) in the genomes of all sequenced hominids (great apes and gibbons), but not in old world monkeys (baboons and macaques), particularly regarding HERV-H (Magiorkinis et al., 2015). HERV-H makes up 88% of all the ERV integrations into the human genome in the last 30 My and became extinct over the past 10 My. HERV-H is a gammaretrovirus, which integrated around 45 to 60 mya and has about 962 copies in the human genome (Chuong et al., 2016). HERV-K, with 10 groups in the clade, only one of which is HML-2, on the other hand, entered the genome of ancestral catarrhines about 32 to 44 mya, after the split from New World monkeys and before the split of hominids from the Old World monkeys (Kim & Han, 2015). The sister lineages of HERV-K in most other catarrhines appear to have become extinct. Most remarkably, the HERV-K HML-2 group in humans is the only HERV-K that has continued to replicate since the origin of the catarrhines (Magiorkinis et al., 2015). HERV-K102 is a member of the bioactive HERV-K HML-2 group and appears to be the only known replication competent member both in vitro and in vivo (Laderoute et al., 2007; Laderoute et al., 2015). Accordingly, since phylogenetic evidence supports an association of HERV-K HML-2 activity with protection against integration of orthoretroviruses (i.e., acquisition), this may help substantiate the notion that modern day HERV-K102 particles, along with expression of proteins from other HML-2 elements, might help prevent HIV-1 acquisition and provide sterilizing immunity (Figure 4).
On the curious origins of HERV-K102 in humans
Somewhat ironically, humans apparently acquired the HERV-K102 defense mechanism from the same source of the modern HIV-1 pandemic strain; namely, chimpanzees, possibly between 500,000 and up to 2 mya (Romano et al., 2006; Subramanian et al., 2011). The Homo-Pan split has been estimated at 6.6 mya (Magiorkinis et al., 2015) or earlier at 7-8 mya (Langergraber et al., 2012).
As mentioned, the HERV-K HML-2 elements originated in primates about 10.3 mya and the CERV-K102 sequence (DQ112149), which is 97% identical to HERV-K102, was estimated to have integrated into chimpanzees at a non-orthologous position about 10 (+/- 3.3) Mya (Romano et al., 2006).
Lentiviruses like HIV-1 may have been active in primates since the divergence of chimpanzees and humans (Katzourakis et al., 2007; Sawyer et al., 2004). Moreover, it has been suggested the ancestor to HIV-1 may have arisen in chimpanzees about 4 mya (Gifford, 2012). Since it has been reported that subsets of chimpanzees with chronic HIV-1 infection showed progression analogous to humans, including greater expression of CD38 in CD8+ HLADR+ T cells (O’Neil et al., 2000), this raises the notion that an HERV-K102 ancestor, as a potential antidote for HIV-1 infection may have been selected through evolution in chimpanzees before it was acquired by humans. The genus Homo arose about 2 mya (Khan N et al., Genome Biol Evol, 2020). Accordingly, it is possible over about a 2 million-year window or longer, the HERV-K102 ancestor may have adapted to an HIV-1 like ancestor lentivirus(es) in chimpanzees prior to its acquisition by humans. Indeed, there is genetic evidence from a 5 amino acid deletion fixed in a human orthologue of tetherin, that humans may have been afflicted by a lentivirus presence long before HIV-1, perhaps about 800,000 years ago, which may have caused changes in innate immune genes in humans (Compton AA et al., Philos Trans R Soc Lond B Biol Sci. 2013). Thus, the phylogenetic evidence raises the notion that HERV-K102 as a replication competent HERV-K HML-2 retrovirus, may have evolved specific mechanisms to limit HIV/lentivirus replication and genome invasion in chimpanzees before being acquired in humans.
Extinct hominins lost HERV-K102 at the orthologous position at 1q22
It is important to note that there is also additional phylogenetic evidence consistent with HERV-K102 protection generally against emerging RNA viruses, and not necessarily limited to orthoretroviruses.
Neanderthals and Denisovans, appear to have lost HERV-K102 at the orthologous chromosome position at 1q22 (Figure 7) and both went extinct. The timing of the last common ancestor was about 0.6 million years ago whereas the Homo species which includes Neanderthals and Denisovans, arose about 2 million years ago (Khan N et al., Genome Biol Evol, 2020). As deletions may be common at the time of insertions, this data raises the possibility that HERV-K102 may have been acquired from chimpanzees by the last common hominin ancestor about 0.6 mya. It should be noted that the earliest modern human remains in Europe so far discovered was about 45,000 years ago (Hajdinjak M et al., Nature, 2021). Thus, at the time of HERV-K102 acquisition the common ancestor would have resided in Africa and this makes it plausible that HERV-K102 may have been acquired from chimpanzees known to live in Africa but not Europe. The finding also by Compton et al. (2013) referenced above implicates an exposure to chimpanzee lentiviruses around the same time, consistent with co-mingling of humans and chimpanzees at that time that HERV-K102 crossed over and into humans.
Figure 7. Evidence for Past Integration and Excision of HERV-K102 at Orthologous Positions in the Neanderthal and Denisovan Genomes.
Direct repeats (gggatg) flank the orthologous HERV-K102 sequence in the human and extinct hominin genomes. The nucleotides marked in red were missing from the Denisovan orthologous position but present in the Neanderthal genome. Most of the HERV-K102 provirus was missing in both extinct hominins with only a few nucleotides corresponding to the ends of the LTRs remaining intact. “n” stands for missing nucleotides. Inquiry was made of the Altai Neanderthal or Denisovan genome at http://bioinf.eva.mpg.de/fetchseq/ on chromosome 1 strand at 155,596,423 to 155,605,644. AF164610 GenBank LTR flanking sequences used: 5’ LTR sequence = AF095801 and 3’ LTR sequence = AF095802.
RNA virus pandemics likely played some role in the demise of the extinct hominins as adaptively introgressed Neanderthal genes in humans frequently feature innate immunity genes against RNA viruses (Enard D & Petro DA, Cell, 2018) or immune pathways against pathogens (Gouy A & Excoffier L, Mol Biol Evol, 2020). As well, innate immune reactions with pathogens affected human evolution between 0.6 and 2 mya (Khan N et al., Genome Biol Evol, 2020). Consistent with this notion of a superior innate defense system of humans versus other interacting hominins was the finding of successful admixture between humans and Neanderthals but only where the female was human. This was deduced by showing mitochondrial DNA carried from the egg (ie., female) in humans contained no introgressed Neanderthal genes (discussed in Bucking R et al., BMC Genomics, 2019). However, it should be appreciated that there may have been other reasons for this one-way admixture aside from innate immunity. Nevertheless, it is tempting to speculate that at least in part, the HERV-K102 protection system may have contributed to the survival of Homo sapiens. If so, one can immediately appreciate the significance of the HERV-K102 protector system against a deadly RNA pandemic virus like SARS-CoV-2, especially the delta variant.
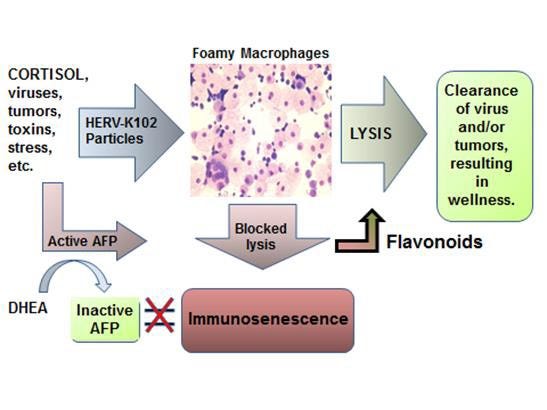
Immunosenescence and HERV-K102 Activity
Before we delve into how SARS-CoV-2 and variants may antagonize the HERV-K102 protector system in Part II (in a separate document), we need to cover the issue of immunosenescence as it affects foamy macrophages.
Immunosenescence has been defined as the failed lytic release of HERV-K102 particles from foamy macrophages (Laderoute M, Discovery Medicine, 2015, 2020). As shown in Figure 8, alpha-fetoprotein (AFP) activity is proposed as the central mediator of immunosenescence. AFP is immunosuppressive, blocks further differentiation and inhibits apoptosis in macrophages (Laderoute M & Pilarski LM, Anticancer Research 1994). In fact, AFP seems to block any incoming signal so the signal to downmodulate the production and release of pro-inflammatory cytokines is also abrogated by AFP. It would seem by the time the 67 kD AFP receptor (Laderoute MP, PhD Thesis, University of Alberta, 1991) is expressed on foamy macrophages, these macrophages are already producing proinflammatory cytokines such as TNF-alpha, IL-6 and IL-1beta. Thus, the resulting foamy macrophages made dysfunctional by AFP binding to its receptor, are both immunosuppressed and paradoxically at the same time, already releasing pro-inflammatory factors.
Figure 8. The New Immunosenescence Paradigm, 2015
In this manner, it is important to note immunosenescence involves both immunosuppression and the UNCONTROLLED release of pro-inflammatory mediators. This clearly distinguishes immunosenescence from a merely pro-inflammatory response. This is vital to understand because the use of anti-inflammatories (which are generally immunosuppressive) will not cure immunosenescence whereas the use of an AFP antagonist may be the only suitable course of action if trying to block immunosenescence. Also the use of immunosuppressive anti-inflammatories could worsen diseases if the etiology involves infectious agents. [For example, cortisol based drugs are dangerous to use in COVID-19 patients during the time where SARS-CoV-2 is actively replicating, but are used later in the inflammatory phase before the onset of cytokine storm.] Thus, while anti-inflammatories are commonly used by allopathic and naturopathic approaches for the treatment of chronic diseases, they do not appear to cure these diseases (they just ameliorate the symptoms). Chronic diseases such a high blood pressure and cardiovascular diseases, cancer, autoimmunity, allergies, and long COVID are all believed to be due to immunosenescence. According to the new immunosenescence paradigm, only AFP antagonists would reverse or prevent immunosenescence and not anti-inflammatories per se. Interestingly anecdotal evidence with ivermectin based prevention protocols has been reported to result in improved health and it is argued elsewhere ivermectin appears to be an AFP antagonist (https://openheart.bmj.com/content/8/1/e001655.responses#ivermectin-may-prevent-and-reverse-immunosenescence-by-antagonizing-alpha-fetoprotein-and-downmodulating-pi3k-akt-mtor-hyperactivity).
In order to prepare for the next pandemic, efforts should be made to research and develop AFP antagonists which would prevent or reverse immunosenescence. Ivermectin, already shown to have beneficial clinical effects for the prevention and treatment (at all stages) of severe COVID-19, should be immediately prioritized to be studied in large well controlled randomized clinical trials for proper determination of efficacy but to include studies on HERV-K102 particle production, genomic copy number, envelope specific antibody and innate T cell responses. We need to address if HERV-K102 activity is needed for recovery against pandemic virus infections. In other studies on innate immunity vaccines, we need to determine if HERV-K102 activity protects against pandemic viruses and to determine how long this lasts.
Dehydroepiandrosterone (DHEA) referred to as the youth hormone is produced by the adrenals and serves to counterbalance the effects of cortisol known as the stress hormone, also produced by the adrenals. With age and/or stress the levels of DHEA diminish while cortisol may marginally increase, and thus, the DHEA/cortisol ratio diminishes with age. DHEA was shown to specifically bind and inactivate AFP establishing it as an AFP antagonist (Laderoute M, Discovery Medicine, 2015). Accordingly, we can now explain how immunosenescence increases with age and/or stress as correlated with the DHEA/cortisol ratio. Indeed, when examined, chronic diseases invariably associate with a poorer DHEA/cortisol ratio when compared with age and sex matched healthy controls. In a study of 125 consecutive patients with COVID-19 related admission to a hospital, the DHEA-S and DHEA-S/cortisol ratio was significantly diminished when comparted with age and sex matched healthy controls. The ratio showed a significant negative correlation with COVID-19 disease severity (Reza VMM et al, Immunoregulation, 2021). Indeed, Tomo S et al., (Endocrine Regulations, September 2021) extols the likely benefits of using DHEA (or preferentially 7-keto-DHEA which cannot be converted to androgens) in the prevention of COVID-19 severity.
In addition to DHEA, flavonoids from the diet also are known AFP antagonists (see more recent work on the flavonoid icaritin for treatment of cancer: Li H et al., BMC Cancer 2021 or for virus infections: Zhang C et al., Oncotarget, 2016). Other potential AFP antagonists as mentioned include ivermectin (see https://openheart.bmj.com/content/8/1/e001655.responses#ivermectin-may-prevent-and-reverse-immunosenescence-by-antagonizing-alpha-fetoprotein-and-downmodulating-pi3k-akt-mtor-hyperactivity) and possibly the anti-androgens. Zinc is known as an AFP antagonist while copper seems to bind and activate AFP.
The reader should recognize that AFP antagonists have been successfully used in the prevention or treatment of COVID-19 by some ‘alternative’ camps in western countries or as adopted in certain other countries. Moreover, there is evidence that these agents may locally halt the pandemic such as in Mexico City and Japan.
What is important to note is that immunosenescence is considered the main risk factor for COVID-19 severity and as will be developed in PART II, SARS-CoV-2 infects both sebocytes and foamy macrophages inducing immunosenescence. This entry into sebocytes and foamy macrophages not only exacerbates or initiates chronic disease but more importantly, SARS-CoV-2 abrogates the protective innate immunity by the HERV-K102 system against pandemic RNA viruses by interfering in particle production/release.
Most significantly however, SARS-CoV-2 upregulates AFP mRNA and protein expression in HuH-7 cells associated with the hyperactivation of the ERBB2/EGFR pathway via PI3K/Akt/mTOR (Appelberg S et al., Emerg Microbes Infect, 2020 & Dr. Ujjwal Neogi, personal communication). This is directly consistent with the notion that SARS-CoV-2 infection in sebocytes/macrophages promotes immunosenescence and blocks the crucial HERV-K102 virus anti-virus response.
Summary of PART I
We have learned that HERV-K102 is a protector foamy retrovirus of humans located on chromosome 1 at 1q22 and that it appears to play an important role in defending humans against pandemic RNA viruses such as HIV-1. It may even have played a role in human survival over the ages. However, traditional approaches to research have failed to identify and thus establish the importance of HERV-K102 particles in host defense, especially against pandemics. There may be constitutive production and lytic release of HERV-K102 particles in sebocytes of sebaceous glands which line mucosal tissues providing the first line of defense against emergent RNA viruses.
Now that we understand what immunosenescence is, we will be able to appreciate how SARS-CoV-2 may promote immunosenescence once it enters sebocytes/foamy macrophages and why this can be so deadly for the host (ie., with the delta variant). As will be discussed in Part II, SARS-CoV-2 uses basignin (BSG/CD147) as a universal receptor for entry which allows it to target the foam generating cells; the sebocytes and macrophages. Of course antibody dependent enhancement (ADE) of SARS-CoV-2 infection into these cells via Fc receptors can be a grave concern when the variant is delta. On the other hand, it could be a god-send for the omicron variant which hopefully will quickly transmit and infect to set up herd INNATE immunity and help us reach endemic status. Stay tuned for upcoming blogs on ADE and why INNATE immunity vaccines may be more appropriate for emerging or pandemic RNA viruses than adaptive immunity ones.









Great question Helen. While not specifically tested, presumably it might? Both are enveloped viruses. However, your HERV-K102 is constantly regenerated so the idea is that gargling is a "good thing". For more information on how to better protect yourself (in addition to the FLCCC protocols) see my substack article on the H.E.R.V.K. Way to Keep SARS-CoV-2 Coronavirus at Bay. Also for prevention, I would not use melatonin above 2 mg/night as it is immunosuppressive. Ditto for turmeric being very immunosuppressive. Note this is a generic recommendation and should not be considered as medical advice. Consult with your health care practitioner. Yes I have been trying to help everyone since March 5, 2020 (before the pandemic was declared), with the release of the H.E.R.V.K. Way to Keep SARS-CoV-2 Coronavirus at Bay protocol [initially posted on LinkedIn but account suspended October 2021 and thus, no longer available on LinkedIn]. Another important point, stay away from plant based protein and use whey protein based supplements as there is an insufficient lysine to arginine ratio in plant proteins as well as diminished tryptophan and tyrosine which imbalances the ability of macrophages to make HERV-K102 particles.
I was ostracized from LinkedIn late October 2021 for posting data consistent with the notion that delta may have emerged in different regions of the world associated with administration of the second COVID-19 vaccine dose in about 50% of the over 50 age group. By this time I had built up a following of about 8000 scientists/clinicians/health-care workers/interested parties. To let them know that I am now writing on substack (hervk102@substack.com) would you be so kind to share Part I and Part II on LinkedIn (as well as other social media sites) on my behalf. It would be so much appreciated. Reviews, discourse and opinions on the HERV-K102 (viral anti-viral) innate protector system against pandemic RNA viruses are also welcomed. Most people are simply unaware of the potential importance of HERV-K102 in fighting pandemics. I thank you in advance from the bottom of my heart.