The Day 10 Post Symptom Onset Window for Seroconversion in COVID-19 Patients
March 12, 2022; Vs. 2.
In my last post, I spoke about how if the HERV-K102 particles and antibody to HERV-K102 envelope have not cleared or inactivated SARS-CoV-2 by around 10 days, then there may be a higher risk of seroconversion. Most people report that seroconversion and neutralizing antibodies (NAbs) correlate with COVID-19 severity (Post N et al., PLoS One, December 2020, a review of 150 studies).
Here is some supporting documentation for this notion. This work is from To KK et al, Lancet Infect Dis, 2020 and data is from January and February 2020, so this is very early in the pandemic. It is necessary to examine what happens without the influence of prior exposures which is why it is important to look at the start of the pandemic.
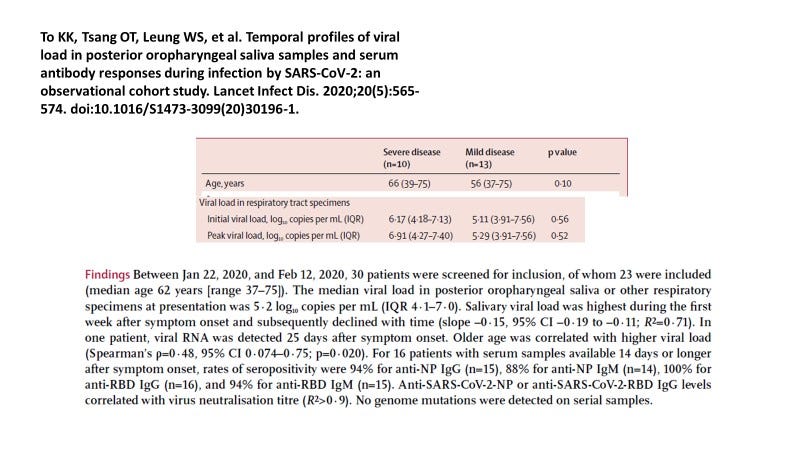
Figure 1. Summary of Findings by To KK et al., 2020.
As given in Figure 1 those who developed severe disease had a saliva viral load (VL) of about 6 x 10 (6) per ml whereas those with mild COVID-19 had about 5 x 10 (6) representing a one log (base 10) difference. [The p value was not significant due to low ‘n’].
Figure 2. RBD IgG antibody titres above 1/100 (eg, 1/200) can be considered a true positive by ELISA.
From my own experience I would not consider that any antibody test not diluted at more than 1/ 100 can give a true positive test by ELISA. In Figure 2, To et al (2020) show that the background can be set around 1.1 when the IgG anti-RBD is competed with free RBD. This matches the background that can be set for 93 serum samples from 2018 (pre-pandemic) plus 3 standard deviations at 0.108. The 1/2 max of inhibition of the specific RBD response is at 1/200. Accordingly, I would say a titre over 1/100 could be considered a true positive.
Figure 3. For the most part the decision to seroconvert occurs around day 10 post symptom onset with 1/2 peak RBD IgG around day 15.
So as mentioned in my last post, day 10 after symptom onset seems key to whether the host will seroconvert (Figure 3). You will note both in the mild and severe cases that sometimes seroconversion starts earlier (7-8 days) perhaps related to higher viral loads or even genetics. It is also possible that those with severe immunosenescence (blocking innate immunity) could have an earlier induction of the adaptive arm of immunity. This is not due to pre-exposures to SARS-CoV-2 as these data are from the very start of the pandemic. In others, we see seroconversion at later times. This could be due to lower viral loads, genetics or because the person is immunosuppressed.
Figure 4. The anti-RBD IgG shows a tight correlation with NAb detection [R(2)=0.96].
When the NAb test is performed with live SARS-CoV-2 propagated on and tested for neutralization within VEROE6 (primate but not human) cells, the anti-RBD IgG shows a tight correlation with NAb detection [R(2)=0.96 for anti-RBD while R(2)=0.99 for anti-NP] as demonstrated in Figure 4. The fact that anti-NP provides neutralizing protection against SARS-CoV-2 raises the issue that natural immunity may provide better protection against COVID-19 than that created by vaccination which only employs spike protein. Regardless, this correlation shows that the NAbs at least represent IgG against the spike protein receptor binding domain (RBD).
Figure 5. It takes about 6-7 days to reduce the saliva VL by about 1 log.
Only 1 log discriminates severe from mild COVID-19 cases (6 logs for severe and 5 logs for mild, see Figure 1). To decrease one log takes about 6-7 days as measured in saliva (Figure 5). Thus, it is critical as to ask what happens during these first 6-7 days after symptom onset.
Figure 6. It takes about 6-7 days for HERV-K102 particles to be induced and released from sebocytes or foamy macrophages by lysis (not shown) and the 1/2 max for plasma accumulation is estimated at about 5 x 10 (5) at around 1.75 days.
It was established that there were 2.55 x 10(11) cDNA containing particles per ml of plasma at 84 hours and none at time zero using the ddCt real time PCR method described by Laderoute M et al, AIDS, 2007. This rapid rise in HERV-K102 particles followed a dechallenge (temporary cessation) of St. John’s wort (hypericum) in a patient with chronic fatigue syndrome. Hypericum also inhibited HERV-K102 replication (cDNA accumulation) in vitro in the foamy macrophages and prevented an increase in provirus gene copy number in genomic DNA (data not shown). This strong induction of HERV-K102 particles was rapidly associated with insomnia, fatigue, and brain fog. These signs of strong induction of HERV-K102 were quickly reversed upon resumption of St. John’s wort. While the chronic fatigue patient had used St. John’s wort for about 15 years she now relies on a product called NuSleep from Immune System Management (Ottawa, Ontario). In addition to counteracting insomnia, fatigue and brain fog, NuSleep was designed to also counteract the adverse effects of stress on sleep and poor sleep quality on innate immunity. Long COVID might involve a post-viral syndrome analogous to chronic fatigue syndrome. It should be noted that for viral infections up to 10 (12) particles per ml of plasma have been reported (Laderoute M et al, AIDS, 2007). On the other hand, In patients with HIV-1, the mean particle number per ml was 8,200 and about 70% positive. However the antibodies to HERV-K102 envelope peptides were strongly induced in 70 to 80% of HIV-1 patients. In total 96 % of patients with HIV-1 had antibodies and/or particles showing the innate HERV-K102 host defense system is UNIVERSALLY activated with PANDEMIC RNA VIRUSES.
The natural log (Ln) of 2.55 x 10 (11) at 84 hours was used to calculate the 1/2 max and other estimates of particles per ml of plasma at different times as provided in Table 1.
In Figure 6, it is shown that the 1/2 max levels of plasma HERV-K102 particles is about 5 x 10 (5) HERV-K102 particles and takes about 42 hours or about 1.75 days to reach.
It is not known if this value bears any relationship to the critical saliva levels of SARS-CoV-2 which distinguishes severe COVID-19 cases [10 (6) SARS-CoV-2 particles/ml of saliva] from mild [10 (5) SARS-CoV-2 particles/ml of saliva ] (Figures 1 and 5) . It does seem to be an interesting co-incidence.
Once there is an initial wave of HERV-K102 particle release (first 6 days after exposure to SARS-CoV-2) in the oral and nasopharyngeal mucosa (and as affecting the foamy sebocytes in sebaceous glands, which are specialized foamy macrophages), it seems another wave is necessary to amplify the response systemically as well as substantially activate the innate B cells producing HERV-K102 envelope antibody. HERV-K102 strong induction is associated with insomnia, fatigue and brain fog (see description in the legend to Figure 6). These signs and symptoms are common not only to long COVID (Stephanou M-I et al. Ther Adv Chronic Disease, Feb 17, 2022) but also during SARS-CoV-2 natural infection.
Thus, it is tempting to speculate that around 6 days after the onset of symptoms, there is a flood of HERV-K102 particles in the blood and a strong activation of the innate B cells which produce antibody to HERV-K102 envelope. [BTW, these HERV-K102 envelope specific antibodies might be primarily IgG2 (Tokuyama M et al, J Exp Med, 2021) and not the IgA or IgG1 associated with the SARS-CoV-2 specific antibodies. IgG2 are weak at binding C1q (complement activation), are not recognized by the FCGR3A or FCGR1A Fc receptors and may only weakly bind FCGR2A (Vidarsson G et al., Front Immunol 2014). ] In about 1.75 days thereafter, the levels of HERV-K102 particles in the blood are proposed to reach the 1/2 max levels as shown in Figure 6. So this brings us to 7.75 to 8.75 days. If we add in 6 hours for the antibodies to induce lysis in the virally infected cells and/or 24 hours for the HERV-K102 particles to mediate rapid cell lysis in virally infected cells, this then brings us to 8.75 to 9.75 days. This then is just before the “10 day post symptom onset” deadline (so to speak) which seems to be the critical decision point for seroconversion.
Indirect Evidence for the Significance of the HERV-K102 Protector Response in COVID-19 Patients
As mentioned in my last post, no one has investigated HERV-K102 particles or antibody to HERV-K102 envelope in COVID-19 patients, despite the methodology being described and published in 2007 (Laderoute M et al., AIDS, 2007) and my efforts to draw attention to this with the various public health authorities at the start of the pandemic. However, Dispinseri S et al, (May 2021) may have unwittingly described the induction of the protector innate neutralizing antibodies as an early event in weeks 1 and 2 from the symptom onset followed by a notable upregulation at 3 to 4 weeks which then becomes downregulated with the onset of the SARS-CoV-2 specific antibodies (Figure 7).
Figure 7. Dispinseri S et al, (Nature Communications, May 2021) reported high “NAb” levels in weeks 1-2 and much higher in weeks 3-4 after symptom onset. However they used a pseudovirus for the test that had been cultured in and released from human cells. As well, there was no true IgG to either RBD or S1 and S2 until weeks 5-8 and onward.
It is striking to note that true IgG antibodies to RBD and S1 and S2 were not evident until 5-8 weeks after symptom onset in the Dispinseri (May 2021) study (Figure 7). This is in agreement with Norval MG et al, (Sci Reports, March 10, 2021) where they showed the majority of COVID-19 patients developed IgG anti-RBD between day 35 and 45 (or about 5 to 6.4 weeks) as measured in 101 recovered health care workers.
They claimed the “NAbs” at 1 to 4 weeks cleared the viral particles from nasopharyngeal swabs (NPS) earlier (than those without “NAbs”) , and that those patients unable to produce these “NAbs” were at a much higher risk of dying.
However, Dispinseri et al. reported that for weeks 5-8 (when the IgG antibodies become detectable) the correlation of the “NAbs” to RBD IgG was R (2) =0.561 while R (2) was 0.476 for the S1+S2 IgG. This poor correlation shows what Dispinseri et al were calling “NAbs” were not SARS-CoV-2 SPECIFIC IgG NAbs (see Figure 4 for the expected R(2) of about 0.96). Moreover, since there was little or no evidence of IgG to spike protein epitopes during the first 4 weeks, poor correlations were entirely expected.
Instead, what likely happened, was that an error was made due to the production of the pseudotyped virus in human cells (rather than green monkey VEROE6 primate cells which are the gold standard for the plaque reduction neutralizing test ie. the PRNT for measuring neutralizing antibodies). Recall that virus particles budding from human cells (used here to make the recombinant pseudotyped particles for testing in the neutralization tests), would pick up HERV-K102 envelope, as HERV-K102 envelope is expressed on transformed and virally infected cells. They used specialized cells called Lenti-X HEK (human embryonic kidney) 293T cells where the T stands for the SV40 T antigen which transforms the 293 cells. Additionally the transfections, which may mimic viral infections, may have served to amplify the levels of the HERV-K102 envelope on the cell surface of human cells. HERV-K102 is unique to humans and is not found in green monkeys.
Therefore it is likely it was the HERV-K102 envelope specific INNATE “neutralizing antibodies” that a) allowed for earlier clearance of SARS-COV-2 particles from the NPS and b) if the host failed to make these innate antibodies they were at higher risk of severity and death. For hospitalized COVID-19 patients, patients with severe COVID-19 had higher anti-RBD when compared with mild patients (Moura AD et al., Sci Reports, Sept 3 2021) a relationship that has been consistent since the beginning of the pandemic (Post et al, 2020).
These innate neutralizing antibodies however, require the release of the HERV-K102 particles which would be inhibited in anyone with immunosenescence (Laderoute M, Discovery Medicine, 2015). Those most at risk would be the aged and/or anyone with comorbidities. Even high glucose could prevent the release of the particles by triggering through IGF-1 and its receptor, blocking apoptosis (cell lysis) and thus the lytic release of HERV-K102 particles from the foamy macrophages/sebocytes. Furthermore, macrophages do not express ACE-2 (vasodilating) but express ACE-1 (vasoconstriction) which becomes upregulated with their differentiation. Thus, HERV-K102 particle production is expected to increase blood pressure (until the cells lyse), and hypertension is perhaps the most commonly reported “co-morbidity” factor for risk of COVID-19 severity. In other words persistent hypertension may signify immunosenescence and a higher risk of COVID-19 severity.
As mentioned, the median levels of innate neutralizing antibodies (I-NAbs) continues until the virus specific antibodies get activated when then there is a corresponding decrease in the I-NAbs median levels (Figure 7). In other words the induction of adaptive immunity downregulates innate immunity. This was also seen at the population level with vaccination where one dose favored innate immunity whereas the second dose favored adaptive immunity and the development of spike specific antibodies and neutralizing antibodies (see my previous post). In this other post, the activation of adaptive immunity seemed to inhibit the negative excess all cause mortality (innate immunity heterologous protection) associated with the administration of the first dose.
So, in conclusion there does seem to be an important day 10 after symptom onset in COVID-19 pathogenesis. Should the induced levels of HERV-K102 envelope specific antibodies and/or particles not be sufficient to neutralize SARS-CoV-2 by around day 10, then it seems the host will seroconvert shortly thereafter with a higher risk of severity and death.
Those with pre-existing immunosenescence (see Laderoute M, Discovery Medicine, 2015 and 2020) such as may be evidenced by persistent hypertension (or possibly also high glucose with or without insulin resistance) may jeopardize the ability of sebocytes or induced foamy macrophages to release the protector HERV-K102 particles, placing the host at increased risk of COVID-19 severity. This should serve as a warning to avoid high levels of glucose during a pandemic even if you are otherwise healthy.





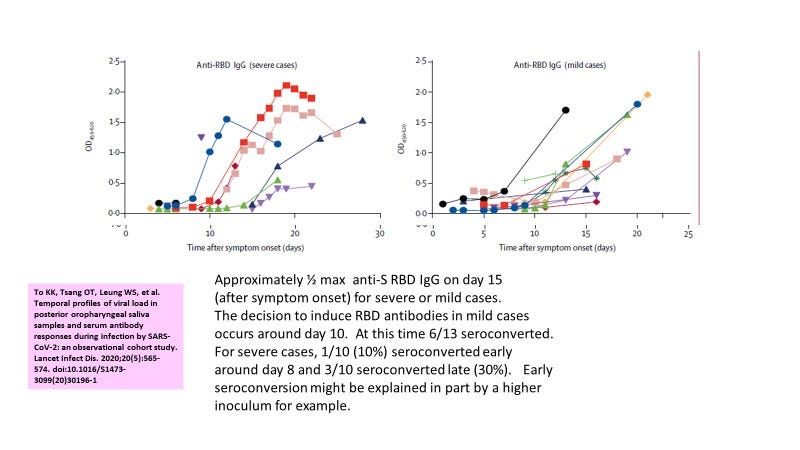




Note that a recent review suggests seroconversion starts around day 12 after COVID-19 symptom onset (Chvatal-Medina M et al., Frontiers in Immunology, April 2021) and where adaptive antibodies to spike protein correlate with severity and not protection.
Another take:
COVID-19 severity is a result of tetanus, flu vaccine-induced allergy to the SARS-CoV-2 virus; Use famotidine, cetirizine, ivermectin to treat
https://vinuarumugham.substack.com/p/covid-19-severity-is-a-result-of?s=w