PHAC/HC COVID-19 VAX Serious Adverse Event Reporting
HACKS to get the SAE into the system where it counts the most.
I was always proud when I worked at Health Canada that in Canada if you had a serious adverse event (SAE) vaccine or otherwise, as a lay person you could report it. The fact that reporting takes time, including obtaining medical documentation, only those doctors getting paid for their submissions could afford to fill out the forms.
As it happens, no doctors are getting paid for reporting. Ergo submissions to SAE databases in Canada are highly unlikely. This leaves it up to the person injured to report, but how?
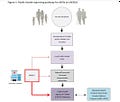
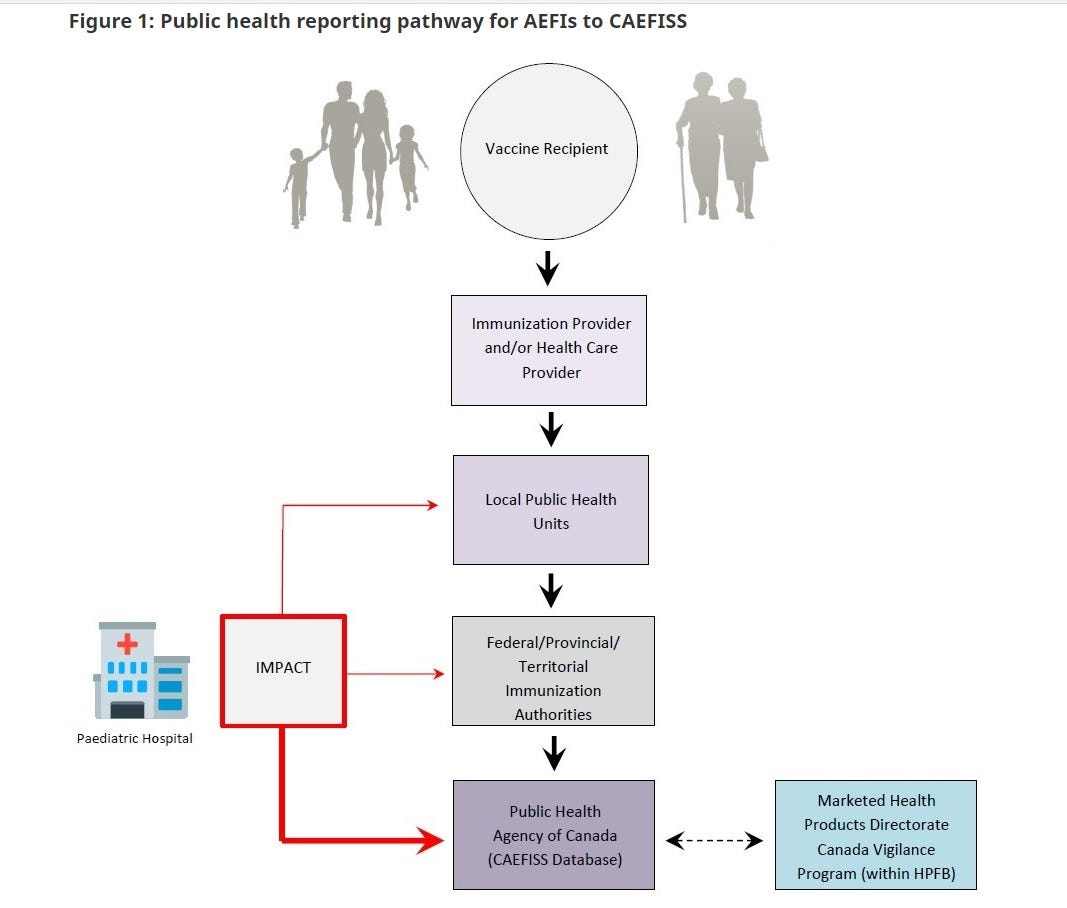
As the above diagram shows, it appears that a lay person reporting the adverse event is no longer tolerated at least for COVID-19 vaccine SAEs. For sure it is denied at the local public health levels, as I recently found out this past fall by a letter requesting that my family doctor report the SAE. Well, I do not have a family doctor and neither did I consult one during my SAE.
So how I got around this foolish system of non-reporting of SAEs is that I reported to the manufacturer, who is required by law to report it to CAEFISS, not only in Canada but around the world. As well, the manufacturer must include an analysis of all SAEs in their Periodic Safety Update Reports (PSURs) to the Marketed Health Products Directorate (MHPD). The MHPD (where on secondment I worked for a year to get a product removed from the market which eventually resulted in a voluntary withdrawal worldwide) has the responsibility of updating the product monograph (warnings, contraindications, % of cases of SAEs etc) and deciding if the product remains on the market.
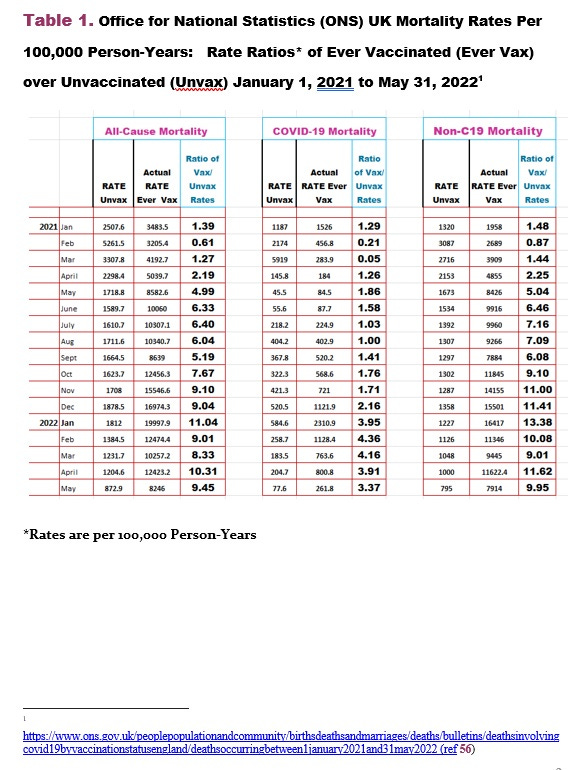
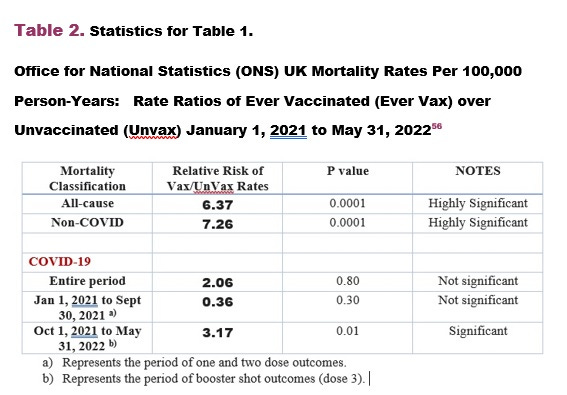
We all know the COVID-19 vaccines should have been removed from the market by the first week of February 2021 because it failed the risk/benefit analysis of all-cause mortality for January 2021 (Tables 1 & 2 below). Even Dr. Peter McCullough said so based on this data in Tables 1 & 2.
Here is the irrefutable data in case you missed it:
And the summary data for overall risk/benefit which shows since the all-cause mortality rate ratios were above 1 and statistically significant (Table 2) means the COVID-19 vaccines (primarily Pfizer BioNTech mRNA in the UK), were neither safe nor effective.
So if you have a serious adverse event following COVID-19 vaccination collect your documents and send the information to the manufacturer. This information will be sent to all regulators in the world where it can provide a more cohesive emerging picture of the lack of safety. Certainly this is much better than not even being registered at the local public health level.
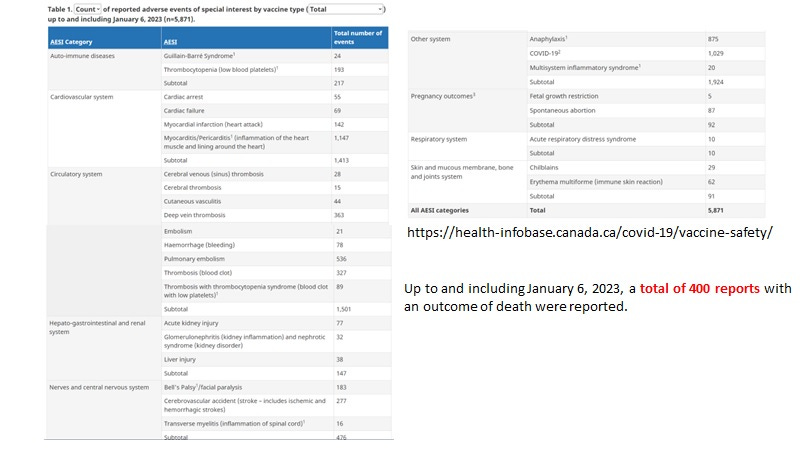
Here is the current summary of the SAEs for COVID-19 vaccines in Canada effective January 20, 2023.
Is it surprising that so few SAEs were reported including deaths? Not really when reporting is actively discouraged.
If we use the UK stats reported in this post:
Then for 37,800,000 UK residents vaccinated with at least 2 doses and having reported an excess of 423,337 non-COVID-19 deaths (over the unvaccinated) means the incidence of vaccine associated deaths (up to end of May 2022) was about 0.01%. Prorated for Canada, with a population of 38,450,000 and 80% vaccinated with at least 2 doses for 30.76 million vaccinated people; at 0.01 % (about 1/10,000), there should have been about 3076 deaths reported to CAEFISS but only 400 were reported (underreporting of about 7.69-fold). However these stats are estimates covering up to the end of May 2022 while the time span is to Jan 20, 2023. So more than likely the number of deaths associated with vaccination in Canada were higher than 3076.
So we need to ensure adverse event reporting by Doctor’s offices are sponsored by HC-PHAC at a reasonable hourly rate, and to allow real time reporting to the feds (CAEFISS) directly so the conglomerate data can be quickly analyzed and acted upon.
Moreover, the manufactures should have been submitting monthly analyses of their PSURs to regulatory authorities by nation and in total (global).



Prof. Patrick Provost of the University of Laval in Quebec City recently published on reasons why it may be difficult to get a physician to report serious adverse events with the COVID-19 vaccines. It can be viewed here:
https://ijvtpr.com/index.php/IJVTPR/article/view/65/170
One of my former colleagues had an interesting adverse event after his "booster" (third shot) and couldn't speak (phonate) for a few days.
I tried to convince him to file "the report", but to no avail.
Then I came up with an idea. He had a medical student that month, so I talked him into having her do the form as a "learning exercise."
I must check back some time and see what outcomes were seen.