How to Distinguish Turbo Cancers
Caused by COVID-19 Vaccine, COVID-19 Infection, Both or Neither
See https://rumble.com/v3ipsi2-dr.-william-makis-september-18-2023-nci-virtual-testimony.html
During a recent NCI/CeNC testimony held virtually on September 18, 2023, Dr. William Makis, an oncologist from Alberta, discussed ‘Turbo Cancers’. Turbo Cancer is a colloquialism to describe those rapidly progressing cancers (often diagnosed at late stages) associated with vaccine breakthrough or temporally associated with either COVID-19 vaccination and/or infection.
There are cases where the person remained unvaccinated but still ended up with turbo cancer subsequent to a COVID-19 infection. So clearly not all cases relate to injections of a gene therapy shot. On the other hand the more doses of mRNA gene therapy shots, the more likely the person will be become infected with a SARS-CoV-2 variant. Presumably this is due to antibody dependent enhancement (ADE) of infection into macrophages in the upper respiratory tract.
Mechanisms causing ADE in the upper respiratory tract (URT) (into sebocytes in sebaceous glands in mucosal tissues) differs from those mediating ADE such as in the lung or lower respiratory tract (LRT) . The former involves FCGR2A on activated sebocytes [Ziegler et al, 2021] while the LRT involves a switch from spike:ACE2 to spike: Basigin (BSG/CD147) [Ren et al, 2021]. Note that BSG [https://www.genecards.org/cgi-bin/carddisp.pl?gene=BSG] aside from operating as an accessory receptor for viruses (SARS-CoV-2, HIV, measles, HCMV etc) and playing a role in foam cell formation, it has been implicated in tumor progression. It is noteworthy that in the URT, BSG is not expressed on the activated sebocytes (foamy macrophages without lipid bodies) but which are nevertheless susceptible to SARS-CoV-2 infection via classical ADE (involves FCGR2A expression in the sebocytes).
Dr. Bernard Massie, an NCI/CeNC Commissioner with a background in molecular biology asked about how to tell if the turbo cancer related to recent vaccination, to recent SARS-CoV-2 infection, both or neither. For example, for myocarditis one examines autopsy heart materials for the spike versus the N protein. If N protein is present it suggests a COVID-19 infection while if absent but the spike protein is present, it implies the vaccine was largely responsible. Those that are negative for both, imply neither the vaccine or infection were involved.
Unlike cases of myocarditis affirmed by autopsy, to date similar analyses of tumor tissue has not been published. It is difficult to say whether spike infestation (vaccines), LNP infestation with or without the SV40 promotor in the Pfizer vaccine [See Image 1] or SARS-COV-2 infection of the tumor tissue would be absolutely required for rapid oncogenesis, or if merely knocking out macrophages and the corresponding potent trained innate immunity [Laderoute MP, submitted] would suffice.
Image 1. SV40 Promoter Sequences Including a Nuclear Localization Signal Not Disclosed to FDA
See McKernan et al, 2023.
Macrophages although long considered the central orchestrator of both innate and adaptive immunity, are primarily associated with innate immunity, the antigen non-specific arm of the immune system involving interferon responses and intrinsic pathogen detection systems. Dendritic cells that are the antigen presenters for adaptive immune responses are derived from a separate set of bone marrow progenitors, called the Common Lymphoid Progenitors (CLPs). Instead macrophages (and monocytes) are derived from the Common Myeloid Progenitors (CMPs) which then generate the Granulocyte-Macrophage Progenitors (GMPs) and the Megakaryocyte-Erythroid Progenitors (MEPs) [Hussein HAM et al, 2023].
So when we talk about monocytes in the blood or about macrophages in the tissues in reality we are talking about innate immunity.
Recently, the expression of S1 protein in CD16+ monocytes in blood was found up to 15 months after COVID-19 infection [Patterson et al, 2023].
First, the blood and URT of the patient should be tested for SARS-CoV-2 (PCR and N protein antigen detection). The person with cancer should indicate details on any COVID-19 infections and/or vaccinations and the dates of cancer diagnosis and all other medical history of relevance including prescriptions and herbal medicines. Antibodies to N protein would suggest a prior (and/or current) infection.
Biopsy or autopsy samples should be prioritized for testing for SARS-COVID-2 proteins, including spike S1 and S2 protein separately, N protein as well as the SV40 promoter/nuclear localization sequences (mRNA versus genomic DNA), and the plasmids noting whether it is tumor cells or myeloid cells that stain positive. Tumor samples commonly contain myeloid cells and where angiogenesis is considered a more lethal attribute [Cheng S et al, 2021]. In situ PCR could be performed to distinguish if it is the tumor, myeloid cells or both that expresses any of the plasmids or SV40 sequences. It is unclear if higher levels of S2 are found in the blood of the mRNA vaccinated than those with natural infection.
There is much discussion about liquid tumor biopsies meaning the mRNA tested in blood is used to determine what kind of cancer and its prognosis [Vong et al, 2021]. The mRNAs from myeloid cells (blood or tissues) are also present. These could be examined for SARS-CoV-2 sequences and the plasmids and other sequences found in the mRNA COVID-19 injections.
In addition DNA from recently lysed cells circulates in blood and its isolation could enrich for genomic DNA corresponding to cells recently killed by the immune system * along with cDNA corresponding to HERV-K102 particles [Laderoute et al, 2007]. This DNA could be scanned for increased integration of HERV-K102 provirus [Laderoute et al, 2015] as well as for the SV40 sequences.
A number of groups have reported on cancer cells and tumor derived exosomes circulating in the blood such as in colorectal cancers (CRCs) [Vafaei et al, 2019]. Apparently these exosomes commonly contain CD63, CD9 and CD81, suggesting they are derived from inflammatory monocytes in the tissues such as from autopsy lung tissue from patients who died from COVID-19 [Delorey et al, 2021]. Presumably in cancer patients at the final stages the inflammatory monocytes might dominate as well.
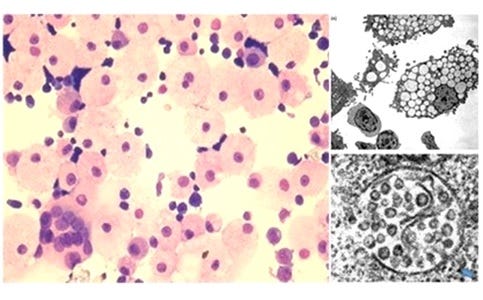
It is unknown if the protector HERV-K102 particles from foamy macrophages (Image 2 similar in size to 100 nm exosomes) contain the CD63, CD9 and CD81 markers. However, since they are from M1-like pro-inflammatory macrophages similar to the Macro_c5 WDR74 COVID-19 recovery myeloid cells [Ren et al, 2021], implies that the HERV-K102 particles likely also express these markers CD63, CD9 and CD81. The finding of myeloid derived exosomes in CRC [Vafaei et al, 2019] implies a role of trained innate immunity involving the lipid-body negative foamy macrophages and HERV-K102 particle production in immuno-surveillance generally against cancers!
Image 2. Protector HERV-K102 particles are produced in foamy macrophages that lack lipid bodies [Laderoute et al, 2015].
There is also evidence that monocyte/macrophage (bone marrow) progenitors may circulate in blood [Hussein et al, 2023] presumably associated with severe disease states. It remains to be determined if their characterization would be useful to discriminate from COVID-19 vaccine versus COVID-19 infection, both or neither.
Tumor tissues depending on what type of cancer, will harbor different types of myeloid cells in the tissues [Cheng et al, 2021]. Thus, it is possible certain myeloid subsets would dominate in mRNA COVID-19 vaccinees versus COVID-19 infected patients which would help distinguish causal assignment.
For example, the myeloid derived suppressor cell (MDSC) found only during COVID-19 infection [Arunachalam et al, 2020] has some similar differentially expressed genes as the C8 myeloid found substantially increased at 2 days after the second dose of the Pfizer COVID-19 vaccine but where the C8 subtype of myeloid cells is associated with heterologous immunity [Arunachalam et al, 2021] (see Image 3).
Image 3. Active Suppression ie, Induction of MDSCs is a feature of COVID-19 Infection Whereas C8 Myeloid Cells (mixed phenotypes consistent with HERV-K102 particle production (IRF1) and trained innate immunity (SPI1 and CEBPB trained immunity enhancers) augmented by interferon gamma) Occur at Higher levels at 2 days following the second dose of the Pfizer vaccine.
From Arunachalam et al, 2020; 2021. However, the authors did not study what happens to the activity of these protector cells with ADE related to vaccination at 30 days after the second dose onward OR IN THOSE WITH SEVERE SARS-COV-2 infection (usually after 10 days) when the IgG1 and IgG3 antibodies to spike protein arise AND immunosuppress the foamy macrophages and cause IMMUNOSENESCENCE. It is known the MDSCs are immunosuppressive and associated with cancer, whereas immunosenescence is associated with loss of tumor and infectious disease immunosurveillance AND CAUSES CHRONIC DISEASES INCLUDING: CANCER.
In summary, it appears we should have the tools to discriminate Turbo Cancers associated with vaccines versus COVID-19 infections. The question is who will fund this research which aims to show that the COVID-19 vaccines are not safe.
_________________________
* Or within the lipid body negative foamy macrophages which release the HERV-K102 particles by lysis (a special type of apoptosis controlled by DNAse I and involving LAMP1-3 proteins in the cytoplasm). One could also look at the DNA in saliva. Since the foamy macrophages producing the HERV-K102 particles are producing and using reverse transcriptase (to enhance particle production), these are the cells of all cells in the body most likely to exhibit reverse transcriptase activity and thus of concern by McKernan and others. FORTUNATELY, IN HUMANS but not rodents, these macrophages (and sebocytes) cannot replicate themselves so they pose no cancer risk.
References
Arunachalam PS, et al. Systems vaccinology of the BNT162b2 mRNA vaccine in humans. Nature. 2021 Aug;596(7872):410-416. doi: 10.1038/s41586-021-03791-x.
Arunachalam PS, et al. Systems biological assessment of immunity to mild versus severe COVID-19 infection in humans. Science. 2020 Sep 4;369(6508):1210-1220. doi: 10.1126/science.abc6261.
Cheng S, Li Z, Gao R, Xing B, Gao Y, Yang Y, Qin S, Zhang L, Ouyang H, Du P, Jiang L, Zhang B, Yang Y, Wang X, Ren X, Bei JX, Hu X, Bu Z, Ji J, Zhang Z. A pan-cancer single-cell transcriptional atlas of tumor infiltrating myeloid cells. Cell. 2021 Feb 4;184(3):792-809.e23. doi: 10.1016/j.cell.2021.01.010.
Delorey TM, et al. COVID-19 tissue atlases reveal SARS-CoV-2 pathology and cellular targets. Nature. 2021 Jul;595(7865):107-113. doi: 10.1038/s41586-021-03570-8.
Dias SSG, Soares VC, Ferreira AC, Sacramento CQ, Fintelman-Rodrigues N, Temerozo JR, Teixeira L, Nunes da Silva MA, Barreto E, Mattos M, de Freitas CS, Azevedo-Quintanilha IG, Manso PPA, Miranda MD, Siqueira MM, Hottz ED, Pão CRR, Bou-Habib DC, Barreto-Vieira DF, Bozza FA, Souza TML, Bozza PT. Lipid droplets fuel SARS-CoV-2 replication and production of inflammatory mediators. PLoS Pathog. 2020 Dec 16;16(12):e1009127. doi: 10.1371/journal.ppat.1009127.
Hussein HAM, Thabet AAA, Mohamed TIA, Elnosary ME, Sobhy A, El-Adly AM, Wardany AA, Bakhiet EK, Afifi MM, Abdulraouf UM, Fathy SM, Sayed NG, Zahran AM. Phenotypical changes of hematopoietic stem and progenitor cells in COVID-19 patients: Correlation with disease status. Cent Eur J Immunol. 2023;48(2):97-110. doi: 10.5114/ceji.2023.129981.
Laderoute MP. Trained immunity involving HERV-K102 activation in foamy macrophages may promote recovery from COVID-19 providing a new innate immunity vaccination paradigm against pandemic RNA viruses. (Submitted).
Laderoute MP, Giulivi A, Larocque L, et al. The replicative activity of human endogenous retrovirus K102 (HERV-K102) with HIV viremia. AIDS. 2007 Nov 30;21(18):2417-24.
Laderoute MP, Larocque LJ, Giulivi A, Diaz-Mitoma F. Further evidence that human endogenous retrovirus K102 is a replication competent foamy virus that may antagonize HIV-1 replication. Open AIDS J. 2015 Dec 7;9:112-22. doi: 10.2174/1874613601509010112.
McKernan, K., Helbert, Y., Kane, L. T., & McLaughlin, S. (2023, April 10). Sequencing of bivalent Moderna and Pfizer mRNA vaccines reveals nanogram to microgram quantities of expression vector dsDNA per dose. https://doi.org/10.31219/osf.io/b9t7m.
Patterson BK, Francisco EB, Yogendra R, Long E, Pise A, Rodrigues H, Hall E, Herrera M, Parikh P, Guevara-Coto J, Triche TJ, Scott P, Hekmati S, Maglinte D, Chang X, Mora-Rodríguez RA, Mora J. Persistence of SARS CoV-2 S1 Protein in CD16+ Monocytes in Post-Acute Sequelae of COVID-19 (PASC) up to 15 Months Post-Infection. Front Immunol. 2022 Jan 10;12:746021. doi: 10.3389/fimmu.2021.746021.
Ren X, Wen W, Fan X, et al. COVID-19 immune features revealed by a large-scale single-cell transcriptome atlas. Cell. 2021 Apr 1;184(7):1895-1913.e19. doi: 10.1016/j.cell.2021.01.053.
Singh N, Singh BA. S2 subunit of SARS-CoV-2 interacts with tumor suppressor protein p53 and BRCA: an in silico study. Transl Oncol. 2020 Oct;13(10):100814. doi: 10.1016/j.tranon.2020.100814.
Vafaei S, Fattahi F, Ebrahimi M, Janani L, Shariftabrizi A, Madjd Z. Common molecular markers between circulating tumor cells and blood exosomes in colorectal cancer: a systematic and analytical review. Cancer Manag Res. 2019 Sep 25;11:8669-8698. doi: 10.2147/CMAR.S219699.
Vong JSL, Ji L, Heung MMS, Cheng SH, Wong J, Lai PBS, Wong VWS, Chan SL, Chan HLY, Jiang P, Chan KCA, Chiu RWK, Lo YMD. Single Cell and Plasma RNA Sequencing for RNA Liquid Biopsy for Hepatocellular Carcinoma. Clin Chem. 2021 Nov 1;67(11):1492-1502. doi: 10.1093/clinchem/hvab116.
Ziegler CGK, Miao VN, Owings AH, et al. Impaired local intrinsic immunity to SARS-CoV-2 infection in severe COVID-19. Cell. 2021 Sep 2;184(18):4713-4733.e22. doi: 10.1016/j.cell.2021.07.023






Who does fund the research that is bad-for-business?
I can't seem to remember.
;-(
Do you have an estimate of how much the research would cost? There might be a crowd funding opportunity if we get others behind it.