Confusion About the Significance of IgG4 Isotype Dominance of Anti-Spike IgG Antibodies Associated with Serial Immunization with COVID-19 Vaccines:
A Good Thing (not the confusion, just the anti-spike dominance of IgG4).
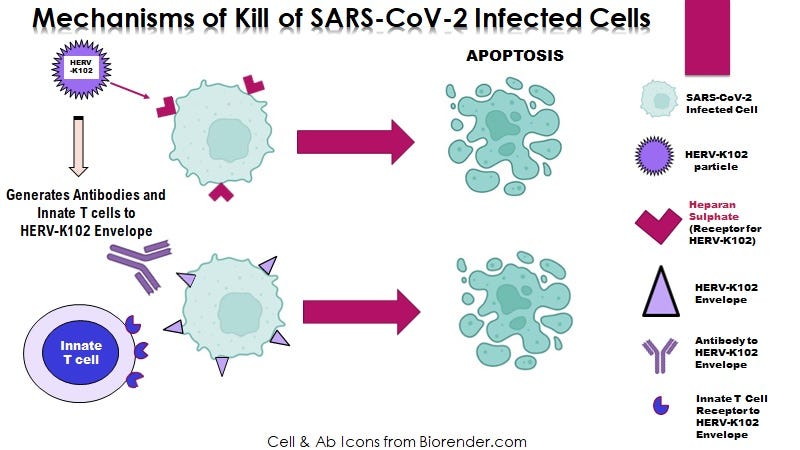
Figure 1. When an enveloped virus buds from human but not non-human cells, the resulting virions contain HERV-K102 envelope which enables INNATE antibodies to HERV-K102 to neutralize and clear the virions. This is very important in the upper respiratory tract for humans to survive RNA virus pandemics.
___________________________________
The following paper created quite a stir amongst clinicians and scientists.
Irrgang P, Gerling J, Kocher K, Lapuente D, Steininger P, Habenicht K, Wytopil M, Beileke S, Schäfer S, Zhong J, Ssebyatika G, Krey T, Falcone V, Schülein C, Peter AS, Nganou-Makamdop K, Hengel H, Held J, Bogdan C, Überla K, Schober K, Winkler TH, Tenbusch M. Class switch towards non-inflammatory, spike-specific IgG4 antibodies after repeated SARS-CoV-2 mRNA vaccination. Sci Immunol. 2022 Dec 22:eade2798. doi: 10.1126/sciimmunol.ade2798.
In this paper, both the FACS-Based antibody assays used for IgG isotyping, and the pVNT omicron pseudotype neutralizing assays would NOT ONLY detect antibodies to spike protein but also antibodies to HERV-K102 envelope protein (Env) (Figure 1). This is because tranduction of human cells induces the HERV-K102 Env at the cell surface as a marker of cells destined for destruction (Figure 1) and get incorporated into the SAR-CoV-2 virions. Only humans have HERV-K102 in their genomes but not other species. So this issue only applies to transduced human cells. In addition since the pVNT assay involved lysis followed by luminometer detection, lysis induced by the HERV-K102 particles in plasma/serum, would falsely elevate the level of “neutralization by antibodies”.
HERV-K102 particles/transcripts are detected in COVID-19 patients [1-4] and are inversely associated with severity [1]. HERV-K102 particles and antibodies to HERV-K102 Env are detected in patients with active viral infections including SARS-CoV-2 [1-4, 5].
So the question emerges is it the anti-spike (ADAPTIVE IMMUNITY-PATHOLOGICAL) or anti-HERV-K102 Env antibodies (INNATE IMMUNITY- PROTECTOR) which become IgG4 with serial vaccination? or both?
Figure 3B of the Irrgang et al paper shows the IgG4 has specificity for RBD but doesn’t technically rule out that some or most of the IgG4 antibodies in Figure 3A may detect the innate antibodies to HERV-K102 Env.
Be that as it may, what if the IgG1/3 to spike protein become largely IgG4 antibodies?
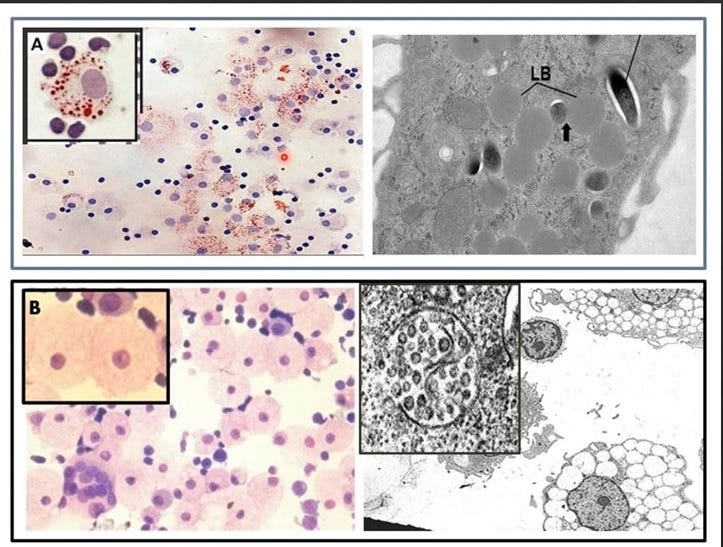
We know from Ziegler et al [6] that in the upper respiratory tract (URT) SARS-CoV-2 preferentially infects the activated sebocytes which are specialized lipid body negative foamy macrophages (LB-FMs) that produce the HERV-K102 protector particles [7]. These cells are constitutively producing HERV-K102 particles and releasing them by lysis into the lumen which then coats the mucosal surface with sebum containing the waxy particles (Figure 2).
Figure 2. Sebocytes have the exact morphology of the LB-FM but CONSTITUTIVELY produce protector HERV-K102 particles in the mucosa (see Figure 3B).
Figure 3. Two types of foamy macrophages in humans.
Upper panel showing both types in response to Mycobacterium tuberculosis exposure in vitro [7] and with the lipid body positive foamy macrophages (LB+FMs) in the insert.
Bottom panel from Laderoute et al, 2007 [4] and Laderoute et al, 2015 [9]. In the insert are shown 2 of the LB-FMS. Only the LB+FMs are oil red o positive as they have cholesterol esters stored in the cytoplasm as lipid bodies. These two types of foamy macrophages are very distinct and sebocytes resemble the LB-FMs producing HERV-K102 particles. It is known that sebocytes phenotypically are LB-FMs [7,10,11] and are immunocompetent [12].
The basal sebocytes express BSG and FCGR2B (inhibitor of FcRs ) but no FCGR2A (the classical receptor mediating antibody dependent enhancement (ADE) of infection of macrophages). So they are refractory to ADE.
To quote from [13]:
“There were 5 types of macrophages distinguished in NPS and macrophages increased in abundance from about 10% in WHO score 1-5 to about 40% in those that subsequently developed severe disease. The IFN responsive macrophages were found to be productively infected with SARS-CoV-2 despite being ACE2 negative. These cells expressed the highest level of FCGR2A but did not express CD16A (FCGR3A) nor FCGR2B, and had the highest level of IFITM3 consistent with IFN responsiveness. Interestingly this was the only macrophage subset that had reduced levels of IFNGR1/2 and surprisingly also the only one that did not express BSG. The trained immunity enhancers SPI1 and CEBPB were expressed by all macrophage subsets except intriguingly not the SARS-CoV-2 infected interferon responsive macrophages. This suggested that SARS-CoV-2 infection may strongly interfere with trained immunity in the nasopharynx (putative sebocytes) more so than in the lungs which uniquely involved the downregulation of BSG. The IFN responsive macrophages were phenotyped as: CD14int CD16-, FCGR2Ahi, B2Mhi, IFITM3hi, CD68int, PML+, NFKB1hi, MX1hi, LAMP1hi, DNASE2lo, IRF1+, STAT1/2+, CTSL+, IDO1+, TLR2+, TLR4+, and S100A9+. Based on the unique expression of ISG20, IFITM1, IFIT1/3, CXCL10/11, GBP1, and highest expression of MX1 in the interferon responsive macrophage subset, these cells most closely resemble the sebocytes described by Nelson et al which were characterized in vitro and in vivo [10, 11]. As well the TLR2 and TLR4 shared pathways for activated sebocytes [12] included IFITM1, CXCL10/11, IDO1, S100A9 and the upregulation of MX1 further substantiating that the macrophages preferentially infected by SARS-CoV-2 may be the sebocytes producing HERV-K102 particles. The high level of LAMP1 and MX1 but low level of DNASE2 is consistent with sebocytes being programmed for cell death but with apoptosis resistance. This would be consistent with SARS-CoV-2 infection of sebocytes causing immunosenescence in the nasopharyngeal mucosa as well as contributing to progression of COVID-19 via upregulation of the CXCL10/11 chemokines, S100A9, IDO1 and/or TLR4.”
These data imply in the URT SARS-CoV-2 enters the activated sebocytes via CLASSICAL ADE, upregulates inflammation, induces apoptosis resistance (ie induces immunosenescence rendering the macrophages dysfunctional and allows for SARS-CoV-2 budding through the cell surface), and knocks out ‘trained immunity’ protection. According to Dias et al [14], the first thing SARS-CoV-2 does upon entering the protective LB-FMs is convert them to the LB+FMs (see Figure 4) which becomes an immunologically privileged site for their replication. Unfortunately the foamy macrophages DO NOT express HERV-K102 Env so they are NOT tagged for destruction.
Figure 4. SARS-CoV-2 Infection of the Protector LB-FMs Converts Them to the Anti-Inflammatory LB+FMs (see Figure 3A for M. tuberculosis) Which Becomes a Factory for the Replication of SARS-CoV-2 As the LB+FMs are an Immunologically Privileged Site (Do not Express HERV-K102 Env)
Note that optimal levels of Vitamin D3 would prevent the ability of SARS-CoV-2 to convert the protector LB-FMs to the non-protector LB+FMs [15]. This would block progression. Indeed, it has been estimated that a Vitamin D3 level of 50 ng/ml or higher would prevent almost all COVID-19 deaths [16]. Vitamin D3 is a much safer and effective means to protect populations against SARS-CoV-2 without generating variants which prolongs the pandemic. Also Vitamin D3 is not toxic like the mRNA vaccines [17] and in fact, promotes health and wellness. Many intracellular pathogens use the strategy of conversion of LB-FMs to LB+FMs [14] including Mycobacterium tuberculosis [8]. If there was no mass vaccination against SARS-CoV-2 the pandemic would have naturally ended in May 2021 [18].
So if the serial vaccination with COVID-19 mRNA vaccines favors IgG4 anti-spike antibodies, these IgG antibodies which ARE detected in the URT (and interestingly not so much after natural infection) [19, 20] would NOT be able to mediate classical FCGR2A ADE in the URT. Therefore, this would stop selection of variants and their transmission to 3rd parties, it would reduce the replication in the URT and thus the transmission to others, and should even reduce the level of viremia and the severity of disease in the individual.
This my friends, would be a good thing.
The reason we have not seen this before is that mass (adaptive immunity) vaccination using a gene therapy vaccine where the antigen persists at high levels had not been experienced before. “This induction of IgG4 antibodies was not observed after homologous or heterologous SARS-CoV-2 vaccination with adenoviral vectors” [Irrgang et al 2022].
In closing, a recent report suggests that one needs >250 BAU/ml (binding antibody units per milliliter of serum standardized by the WHO ; ie., WHO International Reference Standard Units) of neutralizing antibody to prevent SARS-CoV-2 breakthrough infection and severity [21]. What they did not realize was that their methods were detecting the INNATE Protector antibodies to HERV-K102 Env (Figure 1). We know for sure the antibodies to spike are not protective but instead cause the progression to severe COVID-19 disease and death due to ADE infection into the protector LB-FMs (Figure 3B, Figure 4) both in the upper and lower respiratory tracts [7,13].
Only the HERV-K102 protector system can safely protect the human population against an emerging or pandemic RNA virus (Figures 1 & 5) including neutralizing SARS-CoV-2 virions at the entry/transmission points in the URT (Figures 1 & 2).
Figure 5. HERV-K102 particles and innate T and B cell responses defend against SARS-CoV-2 invasion but without recognizing any SARS-CoV-2 specific antigens.
This is a NOVEL VIRUS ANTI-VIRUS MECHANISM of INNATE immunity unique to humans that stands guard (constitutively expressed) at all our mucosal surfaces (which is where the pathogens generally enter) (Figure 2).
It is time to acknowledge the HERV-K102 protector system is key to prevention of COVID-19, is favored by optimal levels of vitamin D, and is absolutely required for prevention of or recovery from, COVID-19 [7,13].
The dynamic, sensitive and reliable methods to confirm this were published in 2007 [4].
References
Desingu PA, Nagarajan K. Unveiling HERV-K113-ENV as SARS-CoV-2 severity admissible biomarker by mining transcriptome data. J Med Virol. 2022 Sep 16:10.1002/jmv.28149. doi: 10.1002/jmv.28149.
Guo Y, Yang C, Liu Y, et al. High expression of HERV-K (HML-2) might stimulate interferon in COVID-19 patients. Viruses. 2022 May 7;14(5):996. doi: 10.3390/v14050996.
Temerozo JR, Fintelman-Rodrigues N, Dos Santos MC, et al. Human endogenous retrovirus K in the respiratory tract is associated with COVID-19 physiopathology. Microbiome. 2022 Apr 22;10(1):65. doi: 10.1186/s40168-022-01260-9.
Laderoute MP, Giulivi A, Larocque L, et al. The replicative activity of human endogenous retrovirus K102 (HERV-K102) with HIV viremia. AIDS. 2007 Nov 30;21(18):2417-24.
Apostolou E, Rizwan M, Moustardas P, et al. Saliva antibody-fingerprint of reactivated latent viruses after mild/asymptomatic COVID-19 is unique in patients with myalgic-encephalomyelitis/chronic fatigue syndrome. Front Immunol. 2022 Oct 20;13:949787. doi: 10.3389/fimmu.2022.949787.
Ziegler CGK, Miao VN, Owings AH, et al. Impaired local intrinsic immunity to SARS-CoV-2 infection in severe COVID-19. Cell. 2021 Sep 2;184(18):4713-4733.e22. doi: 10.1016/j.cell.2021.07.023.
Laderoute MP. Controversies Concerning the Immunology of the COVID-19 Adaptive Immunity Vaccines. pp149 (255 references) [submitted].
Peyron P, Vaubourgeix J, Poquet Y, et al. Foamy macrophages from tuberculous patients' granulomas constitute a nutrient-rich reservoir for M. tuberculosis persistence. PLoS Pathog. 2008 Nov;4(11):e1000204. doi: 10.1371/journal.ppat.1000204.
Laderoute MP, Larocque LJ, Giulivi A, Diaz-Mitoma F. Further evidence that human endogenous retrovirus K102 is a replication competent foamy virus that may antagonize HIV-1 replication. Open AIDS J. 2015 Dec 7;9:112-22. doi: 10.2174/1874613601509010112.
Nelson AM, Zhao W, Gilliland KL, Zaenglein AL, Liu W, Thiboutot DM. Isotretinoin temporally regulates distinct sets of genes in patient skin. J Invest Dermatol. 2009 Apr;129(4):1038-42. doi: 10.1038/jid.2008.338.
Nelson AM, Zhao W, Gilliland KL, Zaenglein AL, Liu W, Thiboutot DM. Neutrophil gelatinase-associated lipocalin mediates 13-cis retinoic acid-induced apoptosis of human sebaceous gland cells. J Clin Invest. 2008 Apr;118(4):1468-78. doi: 10.1172/JCI33869.
Törőcsik D, Kovács D, Póliska S, et al. Genome wide analysis of TLR1/2- and TLR4-activated SZ95 sebocytes reveals a complex immune-competence and identifies serum amyloid A as a marker for activated sebaceous glands. PLoS One. 2018 Jun 21;13(6):e0198323. doi: 10.1371/journal.pone.0198323.
Laderoute MP. Trained immunity involving HERV-K102 activation may promote recovery from COVID-19 providing a new vaccination paradigm against pandemic RNA viruses. pp 81 with 188 refs (submitted).
Dias SSG, Soares VC, Ferreira AC, et al. Lipid droplets fuel SARS-CoV-2 replication and production of inflammatory mediators. PLoS Pathog. 2020 Dec 16;16(12):e1009127. doi: 10.1371/journal.ppat.1009127.
Oh J, Weng S, Felton SK, et al. 1,25(OH)2 vitamin D inhibits foam cell formation and suppresses macrophage cholesterol uptake in patients with type 2 diabetes mellitus. Circulation. 2009 Aug 25;120(8):687-98. doi: 10.1161/CIRCULATIONAHA.109.856070.
Borsche L, Glauner B, von Mendel J. COVID-19 mortality risk correlates inversely with vitamin D3 status, and a mortality rate close to zero could theoretically be achieved at 50 ng/ml 25(OH)D3: results of a systematic review and meta-analysis. Nutrients. 2021 Oct 14;13(10):3596. doi: 10.3390/nu13103596.
Seneff S, Nigh G, Kyriakopoulos AM, McCullough PA. Innate immune suppression by SARS-CoV-2 mRNA vaccinations: The role of G-quadruplexes, exosomes, and microRNAs. Food Chem Toxicol. 2022 Apr 15;164:113008. doi: 10.1016/j.fct.2022.113008.
Kistler KE, Huddleston J, Bedford T. Rapid and parallel adaptive mutations in spike S1 drive clade success in SARS-CoV-2. Cell Host Microbe. 2022 Apr 13;30(4):545-555.e4. doi: 10.1016/j.chom.2022.03.018.
Guerrieri M, Francavilla B, Fiorelli D, et al. Nasal and salivary mucosal humoral immune response elicited by mRNA BNT162b2 COVID-19 vaccine compared to SARS-CoV-2 natural infection. Vaccines (Basel). 2021 Dec 18;9(12):1499. doi: 10.3390/vaccines9121499.
Aksyuk AA, Bansal H, Wilkins D, et al. AZD1222-induced nasal antibody responses are shaped by prior SARS-CoV-2 infection and correlate with virologic outcomes in breakthrough infection. Cell Rep Med. 2022 Dec 15:100882. doi: 10.1016/j.xcrm.2022.100882.
Piñana JL, Vazquez L, Calabuig M, et al. One-year breakthrough SARS-CoV-2 infection and correlates of protection in fully vaccinated hematological patients. Blood Cancer J. 2023 Jan 5;13(1):8. doi: 10.1038/s41408-022-00778-3.





Summary: There are a couple of issues mixed up in the issue of onset of IgG4 dominance with serial C19 vaccination. One is that two of the tests used (FACS Antibody isotyping) and the pVNT for Omicron in the Irrgang P et al, 2022 paper detect the PROTECTOR innate antibodies to HERV-K102 Env and for the latter also the HERV-K102 particles in serum/plasma. The second is the IgG1/3 to spike protein are in the upper respiratory tract (URT) favored by vaccination where they select for and cause the transmission of immune escape variants by ADE which prolongs the pandemic. Their conversion to IgG4 in the URT would reduce this process ending the pandemic sooner and in the host, may reduce the risk of progression to severe disease. So it doesn't necessarily imply it is all bad.
Tokuyama M, Gunn BM, Venkataraman A, Kong Y, Kang I, Rakib T, Townsend MJ, Costenbader KH, Alter G, Iwasaki A. Antibodies against human endogenous retrovirus K102 envelope activate neutrophils in systemic lupus erythematosus. J Exp Med. 2021 Jul 5;218(7):e20191766. doi: 10.1084/jem.20191766. Epub 2021 May 21. Also a sightly different preprint is available at https://doi.org/10.1101/776468 Sept 20, 2019. This paper claims most of the antibody to HERV-K102 is IgG2 with some IgG1.. The complex of soluble HERV-K102 Env (SU domain) with IgG2 antibody activates neutrophils via the FCGR3B (CD16b). SLE plasma (or purified IgG) and recombinant HERV-K102 SU Env form a complex which activates neutrophils: enhances antibody dependent neutrophil phagocytosis (shown by flow cytometry) and increases Neutrophil Extracellular Trap (NET) formation; shown by microscope for fluorescence following Hoeschst staining for extracellular DNA, citrullinated histone 3, and neutrophil elastase.