An Urgent Call for HERV-K102 Research
It's All About Immunosenescence of Macrophages August 23, 2022
Dr. Marian Laderoute is a leading global specialist in the trained innate immunity (heterologous or non-antigen specific) protection mediated by human endogenous retrovirus K102 (HERV-K102). Lipid body negative foamy macrophages (LB-FMs) are pro-inflammatory M1-like foamy macrophages that produce replication competent HERV-K102 particles that are then released by lysis (Figure 1, bottom panel). The released particles are thought to kill virus-infected cells or tumor cells, while in normal cells HERV-K102 integrates increasing the proviral copy number. This ‘arming’ is thought to amplify subsequent HERV-K102 responses and provide innate memory.
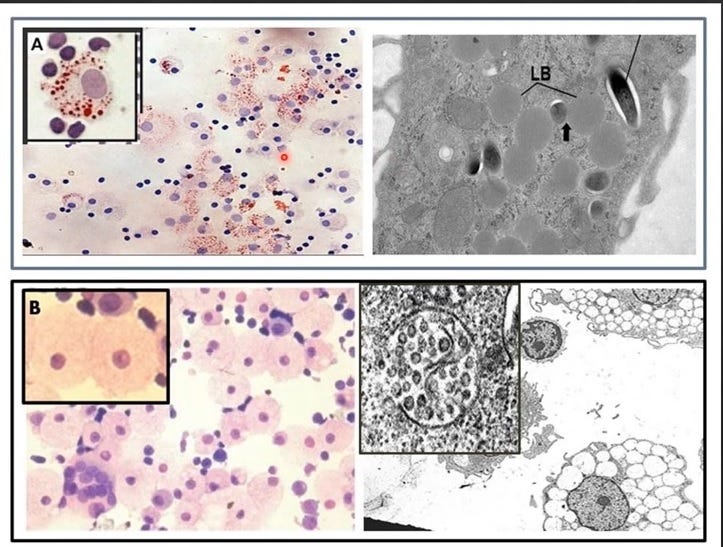
Figure 1. Two Types of Foamy Macrophages in Humans
Top panel: From P. Peyron et al, PLoS Pathogens, 2008 shows two types of foamy macrophages induced in vitro by exposure of human peripheral blood mononuclear cells to Mycobacterium tuberculosis. The lipid body positive foamy macrophages (LB+FMs) (insert A) are Oil Red O (ORO) positive (ORO stains the esterified cholesterol stored as lipid bodies) and the electron micrograph to the right shows M. tuberculosis replicating in the cytoplasm along with the lipid bodies (LB). These LB+FMs are M2-like, anti-inflammatory foamy macrophages which represent a haven for the replication of intracellular pathogens and viruses, including SARS-CoV-2. Indeed, upon infection of monocytes/macrophages, SARS-CoV-2 converts these cells to LB+FMs [Dias SSG et al., PLoS Pathogens, 2020] to favor their replication and to abrogate the protection by the HERV-K102 particles produced in the cells shown in the lower panel.
Bottom panel: From Laderoute MP. F1000 Research, 2018, https://doi.org/10.12688/f1000research.11818.2 and see also Laderoute M et al, AIDS 2007 and Open AIDS J.2015. The lipid body negative foamy macrophages (LB-FMs see insert B) are M1-like, pro-inflammatory foamy macrophages that are highly vacuolated and produce copious amounts of protective HERV-K102 particles which are then released by lysis. Viral infections [Laderoute M, AIDS, 2007] including SARS-CoV-2 induce HERV-K102 [Guo Y et al, Viruses, 2022]. Importantly, HERV-K102 replicative activity was associated with resistance to HIV-1 acquisition as shown in an HIV-1 exposed seronegative cohort [Laderoute M et al, Open AIDS J, 2015]. Ren X et al, Cell 2021 have shown the putative LB-FMs identified as the WDR74 positive macrophages promote recovery from moderate COVID-19 but are diminished associated with progression to severe COVID-19. It is important to note that the progression to severe COVID-19 is associated with the development of adaptive antibodies to spike protein which mediate a switch from spike:ACE-2 (primary receptor) to spike:BSG (secondary receptor). The latter potentiates ADE infection of the WDR74 positive macrophages shown in inset B. Upon entry into the LB-FMs (inset B) SARS-COV-2 converts them into LB+FMs (inset A) which blocks trained innate immunity and at the same time provides a haven for SARS-CoV-2 replication.
HERV-K102 particle release also generates innate T and B cells with reactivity to HERV-K102 envelope. These cells are able to clear virus infected cells and tumors due to the expression of HERV-K HML-2 envelope being expressed on these tumor or virus-transformed cells but not on normal cells. In addition, for enveloped viruses such as HIV-1 and SARS-CoV-2, both examples of RNA pandemic viruses, the budding particles pick up the HERV-K HML-2 envelope (Figure 2), meaning the antibodies to HERV-K102 envelope can neutralize the SARS-CoV-2 and HIV-1 particles. These antibodies to HERV-K envelope are temporary, only last 6 months and appear to be induced by the COVID-19 mRNA vaccines.
Figure 2. Neutralization and Clearance of SARS-CoV-2 Virions by Antibody to HERV-K102 Envelope
These INNATE IMMUNITY antibodies do not recognize viral antigens and thus, cannot provide selection pressure for emergence of immune escape variants. So these innate neutralizing antibodies are ideal for clearing enveloped RNA pandemic viruses like HIV-1 and SARS-CoV-2 irrespective of strain.
It is a myth that the neutralizing antibodies to spike protein induced by COVID-19 vaccination wane by 6 months [Reynolds CJ et al, Science, June 2022]. Instead, it appears that the protection by the HERV-K102 antibodies wane by 6 months and this lack of vaccine effectiveness has led to multiple mRNA vaccine boosting with DIRE consequences.
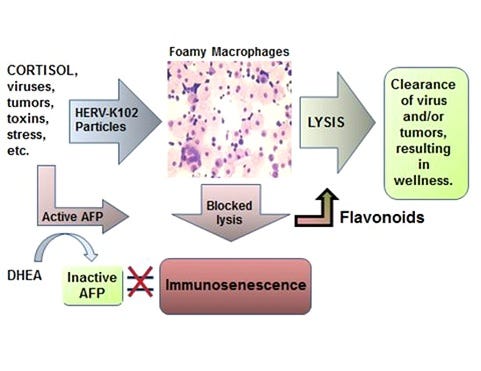
Dr. Laderoute is also a global expert on immunosenescence in macrophages [Laderoute M, Discovery Medicine, 2015, 2020 and see Figure 3] and in the clinical reversion of macrophage immunosenescence with nutraceuticals. Macrophage immunosenescence is causally related to chronic disease including hypertension and cardiovascular diseases [Laderoute M, Discovery Medicine, 2020], cancer, autoimmune diseases, and various metabolic and central nervous system disorders including the dementias. Indeed, correlates of macrophage immunosenescence may include hypertension lasting longer than 7 days and/or elevated CRP. It is becoming clear that SARS-CoV-2 infection induces immunosenescence and those with pre-existing immunosenescence are at increased risk of COVID-19 severity.
Alarmingly, accumulating evidence suggests the COVID-19 vaccines which use or produce spike protein, are very toxic and can lead to death. This problem may be exacerbated in part related to heightened induction of immunosenescence in persons who have neutralizing spike specific antibodies (ie., received the second dose) which favors the spike:BSG secondary receptor interaction over the spike:ACE2 interaction (as described by Ren X et al., Cell, 2021). The spike:BSG interaction seems to cause immunosenescence on its own and may be very dangerous due to lingering spike protein produced for at lease 60 days from the mRNA COVID-19 vaccine. This may have led to an excess of non-COVID-19 deaths (344,0000) noted in the USA as early as MMWR week 14, 2021 (about April 10, 2021) involving cardiovascular disease (20%), cancer (6%) other respiratory illness (3 %), nephritis (3%) etc. [https://the ethicalskeptic.com/?p=67865].
Figure 3. The New Immunosenescence Paradigm for Macrophages 2015 [from Laderoute M, Discovery Medicine, 2015]
The induction of immunosenescence by spike interaction with BSG (spike:BSG secondary receptor) on the activated CD14+CD16+ monocytes (progenitors to the LB-FMs) and/or the activated lipid body negative highly vacuolated foamy macrophages (LB-FMs) may directly induce immunosenescence. The binding of spike to BSG may be favored over the spike/ACE2 primary receptor interaction when there are antibodies and neutralizing antibodies produced to spike protein following the second dose of the mRNA vaccine which block the primary receptor interaction (spike:ACE2). This leaves open the secondary receptor interaction (spike:BSG). This switch in the interaction was described by Ren X et al, Cell 2021 as leading to progression to severe COVID-19 during natural infection with SARS-CoV-2. New evidence now suggests that once spike antibodies and neutralizing antibodies are made in response to the second dose of vaccine (starting around 7-10 days post inoculation), the lingering spike from mRNA vaccines may cause death at any age, due to the induction of immunosenescence. Immunosenescence is no longer restricted to the aged or those with one or more co-morbidities. This helps to explain why there is an increase in hypertension and cardiovascular diseases as well as other chronic diseases.
Later in the third quarter of 2021 when the 25 to 54 year olds had received the second dose, there was a shocking 180 to 200 % increase in excess, non-COVID-19 deaths in the USA [https://rumble.com/v1gocc1-edward-dowd-new-actuarial-report-reaffirms-trail-of-death-caused-by-vaccine.html].
We need to urgently perform clinical research to investigate both spike toxicity (pro-clotting, vasculitis, myocarditis, etc) and the possibility of antibody dependent enhancement (ADE) of immunosenescence in recipients of the second and more doses of mRNA vaccine. The methods for examining the activation of HERV-K102 in monocytes or macrophages (or released HERV-K102 particles in plasma) and for following the 6 month protection by the antibody to HERV-K102 envelope were published in Laderoute M, AIDS, 2007.* We need to test ivermectin along with known nutraceuticals for their ability to reverse immunosenescence to prevent or reverse these vaccine injuries.
For your convenience and to share with others, I am providing a pdf of this post here.
* Laderoute MP, Giulivi A, Larocque L, Bellfoy D, Hou Y, Wu HX, et al. The replicative activity of human endogenous retrovirus K102 (HERV-K102) with HIV viremia. AIDS. 2007 Nov 30;21(18):2417-24.


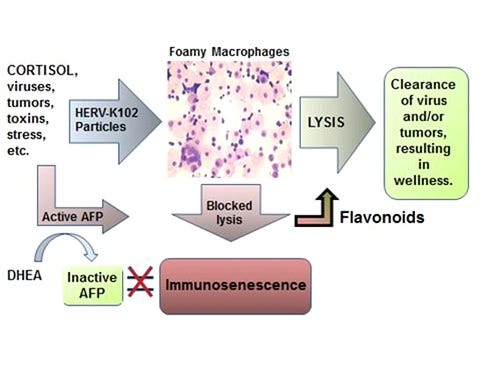
Note: this might be the first time ADE of immunosenescence has been tentatively described. It will need confirmation, however.
https://theethicalskeptic.com/ and @ethicalskeptic on Twitter would be a good resource to explore re: timing of MMWR 14, 2021.
Also note that the same dates show as significant within Australian data.